Polyatomic Ions List Form


What is the Polyatomic Ions List
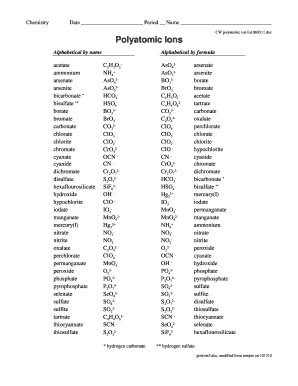
The polyatomic ions list is a compilation of ions that consist of two or more atoms bonded together, carrying a positive or negative charge. These ions play a crucial role in various chemical reactions and are essential in fields such as chemistry and biology. Common examples include sulfate (SO42-), nitrate (NO3-), and ammonium (NH4+). Understanding this list is fundamental for students and professionals working with chemical compounds.
How to Use the Polyatomic Ions List
Using the polyatomic ions list effectively requires familiarity with the structure and charge of each ion. To utilize the list, follow these steps:
- Identify the chemical reaction or compound you are working with.
- Refer to the polyatomic ions list to find the relevant ions involved.
- Use the charges and formulas from the list to balance chemical equations.
- Apply the information in lab settings or during homework assignments.
How to Obtain the Polyatomic Ions List
The polyatomic ions list can be obtained through various educational resources. Here are some methods to access it:
- Download a printable version from educational websites or chemistry textbooks.
- Request a copy from your instructor or educational institution.
- Access online databases or chemistry apps that provide comprehensive lists.
Steps to Complete the Polyatomic Ions List
Completing a polyatomic ions list involves gathering accurate information about each ion. Follow these steps:
- Research common polyatomic ions and their formulas.
- Ensure you include the charge for each ion.
- Organize the ions by their charge or type for easier reference.
- Review your list for accuracy against reliable sources.
Legal Use of the Polyatomic Ions List
The polyatomic ions list can be used legally in educational settings, laboratories, and professional environments. It is important to ensure that any documentation or charts created using this list adhere to academic integrity standards. Proper citation of sources is necessary when using published lists in research or reports.
Examples of Using the Polyatomic Ions List
Here are some practical examples of how the polyatomic ions list can be applied:
- In writing chemical equations, such as when combining sodium (Na+) with sulfate (SO42-) to form sodium sulfate (Na2SO4).
- In laboratory experiments, where precise measurements of compounds containing polyatomic ions are required.
- In educational settings, where students learn to identify and balance chemical reactions involving polyatomic ions.
Quick guide on how to complete polyatomic ions list
Effortlessly Prepare Polyatomic Ions List on Any Device
Digital document management has gained popularity among companies and individuals. It offers an excellent environmentally friendly option to traditional printed and signed documents, as you can locate the appropriate form and safely store it online. airSlate SignNow provides all the tools necessary to create, modify, and eSign your documents swiftly without delays. Manage Polyatomic Ions List on any device using airSlate SignNow's Android or iOS applications and enhance any document-centric process today.
How to Edit and eSign Polyatomic Ions List with Ease
- Obtain Polyatomic Ions List and click Get Form to begin.
- Utilize the tools we offer to fill out your form.
- Emphasize important sections of the documents or redact sensitive information with tools that airSlate SignNow provides specifically for that purpose.
- Create your eSignature using the Sign tool, which takes mere seconds and carries the same legal validity as a conventional handwritten signature.
- Verify all information and click on the Done button to save your modifications.
- Choose your preferred method to send your form, via email, SMS, invitation link, or download it to your computer.
Eliminate concerns about lost or misplaced files, tedious form searching, or mistakes that necessitate printing new document copies. airSlate SignNow fulfills all your document management requirements in just a few clicks from your preferred device. Edit and eSign Polyatomic Ions List and ensure exceptional communication throughout the document preparation process with airSlate SignNow.
Create this form in 5 minutes or less
Create this form in 5 minutes!
How to create an eSignature for the polyatomic ions list
How to create an e-signature for your PDF document online
How to create an e-signature for your PDF document in Google Chrome
The best way to make an electronic signature for signing PDFs in Gmail
How to generate an e-signature from your smart phone
How to generate an electronic signature for a PDF document on iOS
How to generate an e-signature for a PDF file on Android OS
People also ask
-
What is a polyatomic ions list PDF and how can it benefit me?
A polyatomic ions list PDF is a document that provides a comprehensive overview of common polyatomic ions, including their formulas and charges. This resource is beneficial for students, teachers, and professionals in chemistry, as it simplifies the process of understanding complex chemical compounds. With a well-organized PDF, users can reference essential information quickly and efficiently.
-
How can I access a polyatomic ions list PDF through airSlate SignNow?
You can easily create and share a polyatomic ions list PDF using airSlate SignNow by uploading your document to our platform. Our service allows you to eSign and send documents securely, making it simple to collaborate with others. This efficient workflow ensures that your resources, like polyatomic ions lists, are readily available for anyone who needs them.
-
What features does airSlate SignNow offer for document management?
airSlate SignNow offers features such as electronic signatures, document templates, and customizable workflows that enhance document management. You can manage your polyatomic ions list PDF by using our user-friendly interface to organize, send, and track document statuses. These tools help streamline your processes and improve efficiency.
-
Is airSlate SignNow a cost-effective solution for small businesses?
Yes, airSlate SignNow is a cost-effective solution designed for businesses of all sizes, including small enterprises. Our pricing plans are flexible and affordable, allowing you to get the features you need to manage documents like polyatomic ions lists without breaking the bank. Investing in our platform can save you time and resources in the long run.
-
Can I integrate airSlate SignNow with other applications?
Absolutely! AirSlate SignNow integrates seamlessly with a variety of applications, including CRM and project management tools. This means you can easily share and manage your polyatomic ions list PDF alongside other important documents within your existing workflow. These integrations enhance collaboration and streamline your operations.
-
What security measures does airSlate SignNow implement for document protection?
AirSlate SignNow prioritizes security with features like secure cloud storage, encryption, and compliance with industry standards. Your polyatomic ions list PDF and any other sensitive documents are well-protected from unauthorized access. You can trust that your information is safe while using our platform to manage and share important resources.
-
How do I ensure my polyatomic ions list PDF is formatted correctly for sharing?
To ensure your polyatomic ions list PDF is formatted correctly, use our built-in document editor for adjustments before sharing. You can utilize various tools available on airSlate SignNow to organize your content and add necessary annotations. This way, your final output will be clear, professional, and ready for collaboration.
Get more for Polyatomic Ions List
- Concluded that we can not proceed to a proper foreclosure with the state of the title as it is at this form
- Process fee form
- Enclosed herewith please find the settlement documents which have been revised in form
- Enclosed herewith please find check no form
- N a m e debtor form
- N a m e form
- Enclosed herewith please find for your file the following pleadings filed by n a m e in the form
- We have been retained by n a m e in regard to the above referenced loan form
Find out other Polyatomic Ions List
- How Can I eSign Colorado Plumbing PDF
- Can I eSign Hawaii Plumbing PDF
- How Do I eSign Hawaii Plumbing Form
- Can I eSign Hawaii Plumbing Form
- How To eSign Hawaii Plumbing Word
- Help Me With eSign Hawaii Plumbing Document
- How To eSign Hawaii Plumbing Presentation
- How To eSign Maryland Plumbing Document
- How Do I eSign Mississippi Plumbing Word
- Can I eSign New Jersey Plumbing Form
- How Can I eSign Wisconsin Plumbing PPT
- Can I eSign Colorado Real Estate Form
- How To eSign Florida Real Estate Form
- Can I eSign Hawaii Real Estate Word
- How Do I eSign Hawaii Real Estate Word
- How To eSign Hawaii Real Estate Document
- How Do I eSign Hawaii Real Estate Presentation
- How Can I eSign Idaho Real Estate Document
- How Do I eSign Hawaii Sports Document
- Can I eSign Hawaii Sports Presentation