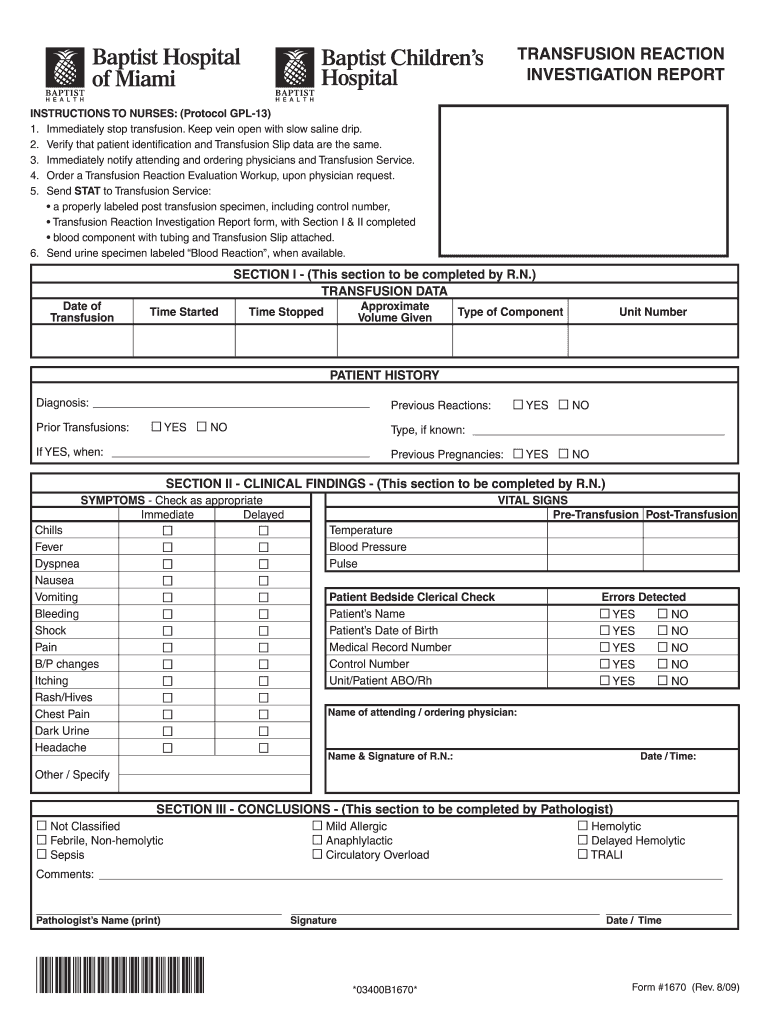
Form 1670 Transfusion Reaction Investigation Report Indd 2009-2026


Key elements of the blood transfusion notes
The blood transfusion notes serve as a critical document in the healthcare process. They include essential information that ensures the safety and effectiveness of the transfusion. Key elements typically found in these notes include:
- Patient Identification: Full name, date of birth, and medical record number.
- Transfusion Type: Details about the type of blood product being transfused, such as red blood cells, platelets, or plasma.
- Indication for Transfusion: The medical reason for the transfusion, including specific diagnoses or conditions.
- Pre-Transfusion Testing: Results of blood type and crossmatch tests, confirming compatibility.
- Consent: Documentation of informed consent obtained from the patient or their representative.
- Administration Details: Time and date of the transfusion, as well as the healthcare provider administering the blood.
- Monitoring Information: Observations made during and after the transfusion, including vital signs and any adverse reactions.
Steps to complete the blood transfusion notes
Completing blood transfusion notes accurately is vital for patient safety and compliance with healthcare regulations. Here are the steps to follow:
- Gather Patient Information: Collect all necessary identification details and medical history relevant to the transfusion.
- Document the Indication: Clearly state the medical reasons for the transfusion, ensuring alignment with clinical guidelines.
- Record Pre-Transfusion Testing: Include results from blood type and compatibility tests, confirming that they meet required standards.
- Obtain Consent: Ensure that informed consent is documented, reflecting the patient's understanding of the procedure and its risks.
- Detail Administration: Note the time and date of the transfusion, along with the name of the administering healthcare professional.
- Monitor and Document: Record vital signs and any reactions during and after the transfusion, noting any complications or concerns.
Legal use of the blood transfusion notes
Blood transfusion notes must comply with various legal and regulatory standards to ensure they are valid and enforceable. Key legal considerations include:
- HIPAA Compliance: Ensure that patient information is protected according to the Health Insurance Portability and Accountability Act.
- Documentation Standards: Follow state and federal guidelines for medical documentation to maintain legal integrity.
- Retention Requirements: Be aware of the legal requirements for retaining medical records, including transfusion notes, for a specified period.
- Informed Consent: Ensure that the consent process is documented to protect against potential legal claims.
How to use the blood transfusion notes format
Using a standardized format for blood transfusion notes enhances clarity and consistency. Here’s how to effectively utilize this format:
- Choose a Template: Select a template that includes all necessary sections, ensuring it meets institutional and regulatory requirements.
- Fill in Patient Details: Accurately enter patient information at the beginning of the notes.
- Follow the Sections: Complete each section in the order specified in the template, ensuring no information is overlooked.
- Review for Accuracy: Double-check all entries for accuracy and completeness before finalizing the document.
- Store Securely: Ensure that completed notes are stored securely in compliance with data protection regulations.
Examples of using the blood transfusion notes
Practical examples of blood transfusion notes can help healthcare professionals understand their application. Here are scenarios illustrating their use:
- Emergency Transfusion: In an emergency situation, a blood transfusion note may document the rapid administration of blood products to a patient with severe anemia.
- Planned Surgery: For a patient scheduled for surgery, the notes may outline the pre-operative transfusions required to optimize hemoglobin levels.
- Adverse Reaction Documentation: If a patient experiences a transfusion reaction, the notes will detail the symptoms, interventions taken, and follow-up care.
Quick guide on how to complete form 1670 transfusion reaction investigation reportindd
Explore the simpler method to handle your Form 1670 Transfusion Reaction Investigation Report indd
The traditional approach to filling out and signifying paperwork consumes an excessive amount of time compared to contemporary document management tools. Previously, you had to search for appropriate social forms, print them, fill in all the details, and dispatch them through the mail. Now, you can locate, fill out, and endorse your Form 1670 Transfusion Reaction Investigation Report indd within a single browser tab using airSlate SignNow. Completing your Form 1670 Transfusion Reaction Investigation Report indd is more straightforward than ever.
Steps to finish your Form 1670 Transfusion Reaction Investigation Report indd with airSlate SignNow
- Navigate to the category page you need and locate your state-specific Form 1670 Transfusion Reaction Investigation Report indd. Alternatively, utilize the search bar.
- Verify that the version of the form is accurate by previewing it.
- Click Get form to enter editing mode.
- Fill in your document with the necessary information using the editing tools.
- Review the added details and select the Sign feature to validate your form.
- Select the most convenient method to create your signature: generate it, draw your signature, or upload an image of it.
- Click DONE to apply the changes.
- Download the document to your device or proceed to Sharing settings to send it digitally.
Efficient online platforms like airSlate SignNow streamline the process of completing and submitting your forms. Give it a try to discover just how long document management and approval processes are truly meant to take. You'll save a considerable amount of time.
Create this form in 5 minutes or less
Create this form in 5 minutes!
How to create an eSignature for the form 1670 transfusion reaction investigation reportindd
How to make an eSignature for the Form 1670 Transfusion Reaction Investigation Reportindd in the online mode
How to generate an electronic signature for the Form 1670 Transfusion Reaction Investigation Reportindd in Chrome
How to make an eSignature for signing the Form 1670 Transfusion Reaction Investigation Reportindd in Gmail
How to make an electronic signature for the Form 1670 Transfusion Reaction Investigation Reportindd from your mobile device
How to make an electronic signature for the Form 1670 Transfusion Reaction Investigation Reportindd on iOS devices
How to make an electronic signature for the Form 1670 Transfusion Reaction Investigation Reportindd on Android OS
People also ask
-
What is the Form 1670 Transfusion Reaction Investigation Report indd?
The Form 1670 Transfusion Reaction Investigation Report indd is a standardized document used in the medical field to report and investigate transfusion reactions. This form ensures that all necessary information is captured for accurate reporting and analysis, helping healthcare providers maintain patient safety.
-
How can airSlate SignNow help with the Form 1670 Transfusion Reaction Investigation Report indd?
airSlate SignNow streamlines the process of filling out and electronically signing the Form 1670 Transfusion Reaction Investigation Report indd. With its user-friendly interface, healthcare professionals can complete the report efficiently, ensuring compliance and reducing administrative burdens.
-
Is there a cost associated with using airSlate SignNow for the Form 1670 Transfusion Reaction Investigation Report indd?
Yes, airSlate SignNow offers a variety of pricing plans to suit different business needs. These plans provide access to features that facilitate the electronic signing and management of documents, including the Form 1670 Transfusion Reaction Investigation Report indd, at a competitive cost.
-
What features does airSlate SignNow offer for managing the Form 1670 Transfusion Reaction Investigation Report indd?
airSlate SignNow includes features such as templates, secure cloud storage, and eSignature capabilities that enhance the management of the Form 1670 Transfusion Reaction Investigation Report indd. These tools help ensure that the document is completed accurately and stored securely for future reference.
-
Can I integrate airSlate SignNow with other software for the Form 1670 Transfusion Reaction Investigation Report indd?
Absolutely! airSlate SignNow offers various integrations with popular software applications, allowing you to seamlessly manage the Form 1670 Transfusion Reaction Investigation Report indd alongside your existing tools. This integration enhances workflow efficiency and minimizes data entry errors.
-
What are the benefits of using airSlate SignNow for the Form 1670 Transfusion Reaction Investigation Report indd?
Using airSlate SignNow for the Form 1670 Transfusion Reaction Investigation Report indd provides numerous benefits, including faster document processing, enhanced security, and improved compliance. The platform's ease of use also allows healthcare professionals to focus more on patient care rather than paperwork.
-
Is airSlate SignNow secure for handling the Form 1670 Transfusion Reaction Investigation Report indd?
Yes, airSlate SignNow prioritizes security, ensuring that all documents, including the Form 1670 Transfusion Reaction Investigation Report indd, are protected through encryption and secure access protocols. This commitment to security helps safeguard sensitive patient information during the signing process.
Get more for Form 1670 Transfusion Reaction Investigation Report indd
- Waiver hunting form
- Waiver and release from liability for minor child for duck hunting form
- Waiver liability house 497427241 form
- Waiver and release from liability for adult for scout function form
- Waiver and release from liability for adult for lodge membership form
- Waiver liability land 497427244 form
- Waiver and release from liability for adult for intramural sports form
- Release minor child 497427246 form
Find out other Form 1670 Transfusion Reaction Investigation Report indd
- How Can I eSignature Wisconsin Orthodontists Word
- How Do I eSignature Arizona Real Estate PDF
- How To eSignature Arkansas Real Estate Document
- How Do I eSignature Oregon Plumbing PPT
- How Do I eSignature Connecticut Real Estate Presentation
- Can I eSignature Arizona Sports PPT
- How Can I eSignature Wisconsin Plumbing Document
- Can I eSignature Massachusetts Real Estate PDF
- How Can I eSignature New Jersey Police Document
- How Can I eSignature New Jersey Real Estate Word
- Can I eSignature Tennessee Police Form
- How Can I eSignature Vermont Police Presentation
- How Do I eSignature Pennsylvania Real Estate Document
- How Do I eSignature Texas Real Estate Document
- How Can I eSignature Colorado Courts PDF
- Can I eSignature Louisiana Courts Document
- How To Electronic signature Arkansas Banking Document
- How Do I Electronic signature California Banking Form
- How Do I eSignature Michigan Courts Document
- Can I eSignature Missouri Courts Document