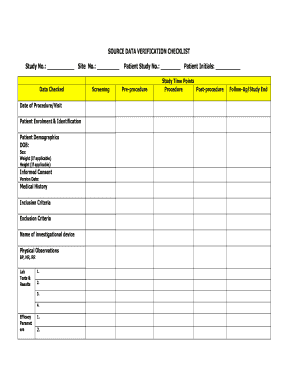
Source Data Verification Checklist Form


What is the Source Data Verification Checklist
The source data verification checklist is a crucial document used to ensure the accuracy and reliability of data collected from various sources. This checklist serves as a systematic guide to verify that the information meets specific standards and requirements. It is commonly utilized in fields such as research, finance, and compliance, where data integrity is paramount. By following this checklist, organizations can minimize errors and maintain high-quality data for decision-making processes.
How to use the Source Data Verification Checklist
Using the source data verification checklist involves a series of steps designed to confirm that all necessary data points are accurate and complete. Start by identifying the data sources and the specific information that needs verification. Next, systematically review each data entry against the original source documentation. This may include cross-referencing with databases, reports, or other records. Ensure that all required fields are filled out and that the data adheres to established formats. Document any discrepancies and take corrective actions as needed.
Key elements of the Source Data Verification Checklist
Several key elements should be included in a source data verification checklist to ensure thoroughness. These elements typically encompass:
- Data Source Identification: Clearly specify where the data originated.
- Data Accuracy: Confirm that the data entries match the original sources.
- Completeness: Ensure that all required data fields are populated.
- Timeliness: Verify that the data is up-to-date and relevant.
- Compliance: Check that the data adheres to applicable regulations and standards.
Steps to complete the Source Data Verification Checklist
Completing the source data verification checklist involves a structured approach. Follow these steps for effective verification:
- Gather Documentation: Collect all necessary source documents and data entries.
- Review Data: Examine each entry for accuracy against the source documents.
- Document Findings: Record any discrepancies or issues encountered during the review.
- Implement Corrections: Make necessary adjustments to rectify any identified errors.
- Finalize Verification: Confirm that all entries meet the checklist criteria before concluding the process.
Legal use of the Source Data Verification Checklist
The legal use of the source data verification checklist is essential in maintaining compliance with various regulations. Organizations must ensure that the data they collect and utilize adheres to relevant laws, such as the Health Insurance Portability and Accountability Act (HIPAA) or the General Data Protection Regulation (GDPR). By employing a verification checklist, businesses can demonstrate due diligence in data handling practices, which can be crucial in legal contexts. This not only protects the organization but also builds trust with clients and stakeholders.
Examples of using the Source Data Verification Checklist
Examples of using the source data verification checklist can be seen across various industries. In healthcare, it may be used to verify patient records against official documentation to ensure accuracy in treatment plans. In finance, organizations might use the checklist to confirm the validity of transaction records before reporting. Additionally, research institutions often rely on this checklist to validate data collected for studies, ensuring that findings are based on accurate and reliable information. Each of these examples highlights the importance of thorough verification in maintaining data integrity.
Quick guide on how to complete source data verification checklist
Complete Source Data Verification Checklist seamlessly on any device
Digital document management has become increasingly favored by businesses and individuals. It serves as an ideal eco-friendly alternative to traditional printed and signed documents, allowing you to find the necessary form and securely store it online. airSlate SignNow equips you with all the resources needed to create, edit, and eSign your documents swiftly without any delays. Manage Source Data Verification Checklist on any device with airSlate SignNow's Android or iOS applications and streamline any document-related process today.
How to modify and eSign Source Data Verification Checklist with ease
- Locate Source Data Verification Checklist and click on Get Form to begin.
- Utilize the tools we offer to complete your document.
- Highlight important sections of your documents or redact sensitive information using tools specifically designed for that purpose by airSlate SignNow.
- Create your signature using the Sign feature, which takes just seconds and carries the same legal weight as a conventional wet ink signature.
- Review the information and click on the Done button to save your updates.
- Choose how you wish to share your form, whether by email, text message (SMS), invitation link, or download it to your computer.
Eliminate concerns about lost or misplaced documents, tedious form searches, or errors that necessitate printing new document copies. airSlate SignNow addresses all your document management needs in a few clicks from your preferred device. Modify and eSign Source Data Verification Checklist and ensure exceptional communication throughout the form preparation process with airSlate SignNow.
Create this form in 5 minutes or less
Create this form in 5 minutes!
How to create an eSignature for the source data verification checklist
How to create an electronic signature for a PDF online
How to create an electronic signature for a PDF in Google Chrome
How to create an e-signature for signing PDFs in Gmail
How to create an e-signature right from your smartphone
How to create an e-signature for a PDF on iOS
How to create an e-signature for a PDF on Android
People also ask
-
What is a source data verification checklist?
A source data verification checklist is a structured document used to ensure the accuracy and completeness of data captured during a project. It helps businesses identify discrepancies and validate source data effectively before further processing. Implementing a checklist can greatly enhance data integrity and accountability.
-
How can airSlate SignNow assist with my source data verification checklist?
airSlate SignNow streamlines the process of creating and managing your source data verification checklist. With its easy-to-use platform, you can automate document workflows, ensuring that every checklist is accessible and securely eSigned. This ultimately increases efficiency in data verification tasks.
-
Is airSlate SignNow a cost-effective solution for managing checklists?
Yes, airSlate SignNow is designed to be a cost-effective solution for businesses of all sizes. By providing tools to create and manage your source data verification checklist online, you save both time and money. Our pricing plans are competitive, ensuring robust features without breaking your budget.
-
What features does airSlate SignNow offer for creating checklists?
airSlate SignNow offers a variety of features for creating checklists, including customizable templates, automated reminders, and secure eSignature options. You can easily manage your source data verification checklist within a user-friendly dashboard, facilitating smooth collaboration among team members.
-
Can I integrate airSlate SignNow with other software for my verification processes?
Absolutely! airSlate SignNow offers seamless integrations with various software applications, enhancing your source data verification checklist management. Popular integrations include CRM systems, project management tools, and more, ensuring your workflows remain efficient and centralized.
-
What are the benefits of using a digital source data verification checklist?
Using a digital source data verification checklist through airSlate SignNow increases accessibility, reduces paperwork, and enhances collaboration. Digital checklists can be easily updated, shared, and stored securely. This helps maintain an organized approach to data verification while ensuring that everyone is on the same page.
-
Is there customer support available for airSlate SignNow users?
Yes, airSlate SignNow provides excellent customer support for all users. Our team is available to assist you with any questions regarding your source data verification checklist or any other features of the platform. We are committed to helping you achieve the best results with our solution.
Get more for Source Data Verification Checklist
- Professional liability questionnaire great american form
- Application for pediatric transplant hepatology training form
- Crisp reporting services hscrcstatemdus form
- 272 alpha drive p form
- Axis student accident and sickness claim form
- Excellus claim form
- Tool 123 template for physicians letter to the child adolescents form
- 2020 patient information amp permanent lifetime signature
Find out other Source Data Verification Checklist
- eSign California Real Estate Business Plan Template Free
- How Can I eSign Arkansas Real Estate Promissory Note Template
- eSign Connecticut Real Estate LLC Operating Agreement Later
- eSign Connecticut Real Estate LLC Operating Agreement Free
- eSign Real Estate Document Florida Online
- eSign Delaware Real Estate Quitclaim Deed Easy
- eSign Hawaii Real Estate Agreement Online
- Help Me With eSign Hawaii Real Estate Letter Of Intent
- eSign Florida Real Estate Residential Lease Agreement Simple
- eSign Florida Real Estate Limited Power Of Attorney Online
- eSign Hawaii Sports RFP Safe
- eSign Hawaii Sports Warranty Deed Myself
- eSign Louisiana Real Estate Last Will And Testament Easy
- eSign Louisiana Real Estate Work Order Now
- eSign Maine Real Estate LLC Operating Agreement Simple
- eSign Maine Real Estate Memorandum Of Understanding Mobile
- How To eSign Michigan Real Estate Business Plan Template
- eSign Minnesota Real Estate Living Will Free
- eSign Massachusetts Real Estate Quitclaim Deed Myself
- eSign Missouri Real Estate Affidavit Of Heirship Simple