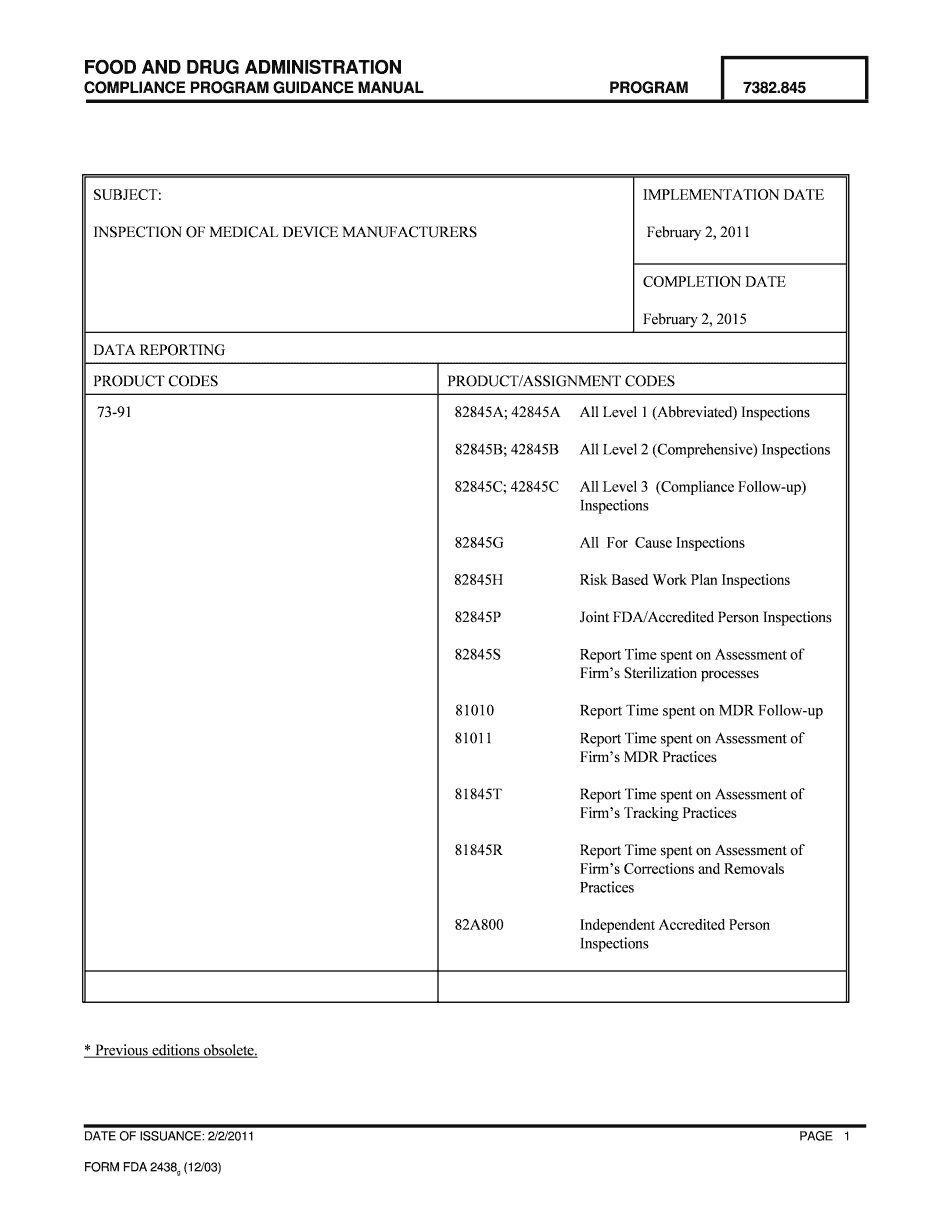
Attachment E CDRH Final Guidance Cover Sheet FDA Form


What is the CDRH cover sheet?
The CDRH cover sheet is a crucial document used in the submission process for medical devices and related products to the FDA. It serves as a summary of the submission, providing essential information about the applicant, the product, and the intended use. The cover sheet is part of the Attachment E to the CDRH Final Guidance, which outlines the requirements for submissions to ensure compliance with FDA regulations. This document is vital for facilitating the review process by clearly presenting key details to FDA reviewers.
Steps to complete the CDRH cover sheet
Completing the CDRH cover sheet involves several important steps to ensure accuracy and compliance. Begin by gathering all necessary information about your product and submission. Follow these steps:
- Provide the applicant's name and contact information.
- Include the product's name, common name, and classification.
- Specify the type of submission, such as a premarket notification (510(k)) or premarket approval (PMA).
- Detail the intended use and indications for the device.
- List any relevant prior submissions or related documents.
Review the completed cover sheet for accuracy before submission, as errors can lead to delays in the review process.
Key elements of the CDRH cover sheet
The CDRH cover sheet contains several key elements that are essential for a successful submission. These include:
- Applicant Information: Name, address, and contact details of the submitting party.
- Device Identification: Product name, common name, and classification information.
- Submission Type: Indication of whether the submission is a 510(k), PMA, or other type.
- Intended Use: Clear description of the device's purpose and indications.
- Regulatory History: Information on any previous submissions related to the device.
Including these elements accurately helps ensure that the FDA can efficiently process the submission.
Legal use of the CDRH cover sheet
The CDRH cover sheet is legally binding when submitted as part of an official application to the FDA. It must comply with the relevant regulations set forth by the FDA to ensure that all information provided is truthful and accurate. Misrepresentation or failure to disclose necessary information can lead to penalties, including rejection of the submission or legal repercussions. Therefore, it is essential to understand the legal implications of the information presented on the cover sheet.
Form submission methods for the CDRH cover sheet
The CDRH cover sheet can be submitted through various methods, depending on the type of submission. Common submission methods include:
- Online Submission: Many submissions can be made electronically through the FDA's electronic submission gateway.
- Mail: Physical copies of the cover sheet and accompanying documents can be sent via postal service.
- In-Person: Some applicants may choose to deliver their submissions directly to the FDA office.
Each method has specific requirements and timelines, so it is important to choose the most appropriate option for your submission.
Examples of using the CDRH cover sheet
Examples of using the CDRH cover sheet can provide clarity on its application. For instance, a manufacturer submitting a new medical device for approval would fill out the cover sheet with details about the device, including its classification and intended use. Another example includes a company seeking to modify an existing device, where the cover sheet would reflect the changes and provide context for the FDA review. These examples illustrate how the cover sheet is tailored to the specific submission type and helps facilitate the regulatory process.
Quick guide on how to complete attachment e cdrh final guidance cover sheet fda
Complete Attachment E CDRH Final Guidance Cover Sheet FDA effortlessly on any device
Online document management has gained popularity among organizations and individuals alike. It offers an ideal eco-friendly alternative to traditional printed and signed forms, allowing you to access the necessary documents and securely save them online. airSlate SignNow provides you with all the resources required to create, modify, and electronically sign your documents quickly without delays. Manage Attachment E CDRH Final Guidance Cover Sheet FDA on any device using airSlate SignNow's Android or iOS applications and enhance your document-related processes today.
How to modify and electronically sign Attachment E CDRH Final Guidance Cover Sheet FDA with ease
- Find Attachment E CDRH Final Guidance Cover Sheet FDA and then click Get Form to begin.
- Utilize the tools we offer to complete your document.
- Emphasize important sections of your documents or redact sensitive data with tools provided by airSlate SignNow specifically for these tasks.
- Generate your electronic signature using the Sign tool, which takes only seconds and holds the same legal validity as a conventional handwritten signature.
- Review the details and then click on the Done button to save your changes.
- Choose how you would like to send your form, whether by email, SMS, or invitation link, or download it to your computer.
Say goodbye to lost or misplaced documents, tiring searches for forms, or mistakes that require new document copies. airSlate SignNow fulfills all your document management needs in just a few clicks from any device you prefer. Modify and electronically sign Attachment E CDRH Final Guidance Cover Sheet FDA and ensure excellent communication at every stage of your form preparation process with airSlate SignNow.
Create this form in 5 minutes or less
Create this form in 5 minutes!
How to create an eSignature for the attachment e cdrh final guidance cover sheet fda
How to make an electronic signature for the Attachment E Cdrh Final Guidance Cover Sheet Fda in the online mode
How to make an eSignature for the Attachment E Cdrh Final Guidance Cover Sheet Fda in Chrome
How to create an eSignature for signing the Attachment E Cdrh Final Guidance Cover Sheet Fda in Gmail
How to generate an eSignature for the Attachment E Cdrh Final Guidance Cover Sheet Fda straight from your smart phone
How to create an eSignature for the Attachment E Cdrh Final Guidance Cover Sheet Fda on iOS devices
How to create an electronic signature for the Attachment E Cdrh Final Guidance Cover Sheet Fda on Android devices
People also ask
-
What is the Attachment E CDRH Final Guidance Cover Sheet FDA and why is it important?
The Attachment E CDRH Final Guidance Cover Sheet FDA is a crucial document required for submitting medical device applications to the FDA. It provides essential information about the application and helps streamline the review process. Understanding this cover sheet is vital for ensuring compliance and expediting approval.
-
How can airSlate SignNow assist with completing the Attachment E CDRH Final Guidance Cover Sheet FDA?
airSlate SignNow simplifies the process of completing the Attachment E CDRH Final Guidance Cover Sheet FDA by offering customizable templates and easy document editing features. Our platform allows you to fill out the necessary information quickly and securely. With eSignature capabilities, you can ensure that all parties can sign off on the document efficiently.
-
What are the pricing plans for using airSlate SignNow for the Attachment E CDRH Final Guidance Cover Sheet FDA?
airSlate SignNow offers competitive pricing plans that cater to various business needs, including options for teams and enterprises. Our plans include features that facilitate the completion of the Attachment E CDRH Final Guidance Cover Sheet FDA at a cost-effective rate. You can choose a plan that best suits your organization's size and budget.
-
What features does airSlate SignNow offer for managing the Attachment E CDRH Final Guidance Cover Sheet FDA?
airSlate SignNow provides a range of features designed to manage documents efficiently, including eSigning, templates for the Attachment E CDRH Final Guidance Cover Sheet FDA, and secure cloud storage. These features ensure that your documents are easily accessible, editable, and shareable with colleagues and stakeholders. This leads to a more streamlined document management process.
-
Is airSlate SignNow compliant with FDA regulations when handling the Attachment E CDRH Final Guidance Cover Sheet FDA?
Yes, airSlate SignNow is compliant with FDA regulations, making it a reliable choice for managing the Attachment E CDRH Final Guidance Cover Sheet FDA. Our platform adheres to industry standards for security and data protection, ensuring that your sensitive documents are handled safely. This compliance helps you maintain the integrity of your submissions to the FDA.
-
Can I integrate airSlate SignNow with other tools for managing the Attachment E CDRH Final Guidance Cover Sheet FDA?
Absolutely! airSlate SignNow integrates seamlessly with various applications such as Google Drive, Salesforce, and others, making it easier to manage the Attachment E CDRH Final Guidance Cover Sheet FDA within your existing workflows. These integrations enhance productivity and allow for a more cohesive document management experience.
-
How does airSlate SignNow enhance collaboration on the Attachment E CDRH Final Guidance Cover Sheet FDA?
airSlate SignNow enhances collaboration by allowing multiple users to access, edit, and sign the Attachment E CDRH Final Guidance Cover Sheet FDA in real-time. You can invite team members to collaborate on the document, ensuring that all relevant input is gathered efficiently. This collaborative approach helps in creating a comprehensive and accurate submission.
Get more for Attachment E CDRH Final Guidance Cover Sheet FDA
- Application for manufactured home community state of michigan michigan form
- Open public records act opra form
- Short answer questions chapter 1 form
- Structure contract template form
- Stucco contract template form
- Stud dog breed contract template form
- Stud dog contract template form
- Stud fee contract template form
Find out other Attachment E CDRH Final Guidance Cover Sheet FDA
- Electronic signature Michigan Email Cover Letter Template Free
- Electronic signature Delaware Termination Letter Template Now
- How Can I Electronic signature Washington Employee Performance Review Template
- Electronic signature Florida Independent Contractor Agreement Template Now
- Electronic signature Michigan Independent Contractor Agreement Template Now
- Electronic signature Oregon Independent Contractor Agreement Template Computer
- Electronic signature Texas Independent Contractor Agreement Template Later
- Electronic signature Florida Employee Referral Form Secure
- How To Electronic signature Florida CV Form Template
- Electronic signature Mississippi CV Form Template Easy
- Electronic signature Ohio CV Form Template Safe
- Electronic signature Nevada Employee Reference Request Mobile
- How To Electronic signature Washington Employee Reference Request
- Electronic signature New York Working Time Control Form Easy
- How To Electronic signature Kansas Software Development Proposal Template
- Electronic signature Utah Mobile App Design Proposal Template Fast
- Electronic signature Nevada Software Development Agreement Template Free
- Electronic signature New York Operating Agreement Safe
- How To eSignature Indiana Reseller Agreement
- Electronic signature Delaware Joint Venture Agreement Template Free