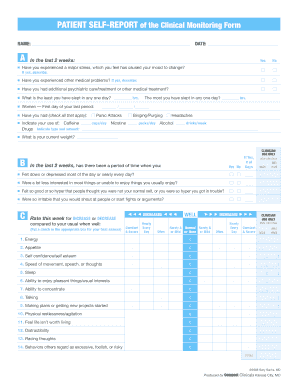
Clinical Monitoring Form


What is the Clinical Monitoring Form
The clinical monitoring form is a vital document used in various healthcare settings to track patient progress, treatment efficacy, and adherence to clinical protocols. This form serves as a record for healthcare professionals to document observations, interventions, and outcomes related to patient care. It is commonly utilized in clinical trials, research studies, and routine patient management to ensure that all necessary data is collected systematically and accurately.
How to Use the Clinical Monitoring Form
Using the clinical monitoring form effectively requires attention to detail and adherence to established guidelines. Start by ensuring that all relevant patient information is accurately filled out, including demographics, medical history, and specific clinical indicators. Healthcare providers should regularly update the form during patient visits or clinical assessments to reflect any changes in the patient's condition or treatment plan. This ongoing documentation helps maintain a comprehensive overview of the patient's journey and supports informed decision-making.
Steps to Complete the Clinical Monitoring Form
Completing the clinical monitoring form involves several key steps:
- Gather necessary patient information, including personal details and medical history.
- Document clinical observations and assessments, ensuring accuracy and clarity.
- Include any interventions or treatments administered during the monitoring period.
- Review the completed form for completeness and accuracy before submission.
- Store the form securely, maintaining confidentiality and compliance with relevant regulations.
Legal Use of the Clinical Monitoring Form
The clinical monitoring form must be used in accordance with legal and regulatory standards to ensure its validity and compliance. This includes adhering to privacy laws such as HIPAA, which protects patient information, and ensuring that the form is filled out accurately to avoid potential legal issues. Proper documentation can serve as a legal record in case of disputes or audits, highlighting the importance of thorough and precise entries in the clinical monitoring form.
Key Elements of the Clinical Monitoring Form
Several key elements are essential for the clinical monitoring form to be effective:
- Patient Identification: Unique identifiers such as patient ID numbers and contact information.
- Clinical Data: Relevant medical history, current medications, and treatment protocols.
- Observations: Detailed notes on patient assessments, symptoms, and responses to treatment.
- Signatures: Required signatures from healthcare providers to validate the entries made in the form.
Examples of Using the Clinical Monitoring Form
Clinical monitoring forms can be employed in various scenarios, including:
- Tracking patient progress in clinical trials to evaluate the effectiveness of new treatments.
- Documenting routine patient visits in outpatient settings to monitor chronic conditions.
- Recording adverse events or complications during treatment to ensure patient safety.
Quick guide on how to complete clinical monitoring form
Effortlessly Prepare Clinical Monitoring Form on Any Device
Online document administration has become increasingly popular among businesses and individuals. It serves as an ideal environmentally friendly alternative to conventional printed and signed papers, allowing you to obtain the necessary form and securely store it online. airSlate SignNow equips you with all the tools required to create, modify, and electronically sign your documents swiftly without complications. Manage Clinical Monitoring Form on any platform with airSlate SignNow's Android or iOS applications and enhance any document-oriented process today.
Efficiently Modify and Electronically Sign Clinical Monitoring Form with Ease
- Find Clinical Monitoring Form and click on Get Form to begin.
- Utilize the tools we provide to fill out your form.
- Emphasize relevant sections of the documents or obscure sensitive information using tools that airSlate SignNow specifically provides for this function.
- Generate your eSignature with the Sign tool, which takes seconds and holds the same legal significance as a traditional handwritten signature.
- Review the details and click on the Done button to preserve your changes.
- Decide how you wish to send your form, via email, text message (SMS), or invite link, or download it to your computer.
Eliminate concerns about lost or misplaced documents, tedious form searching, or mistakes that require printing new sets of papers. airSlate SignNow meets your document management needs in just a few clicks from any device you prefer. Modify and electronically sign Clinical Monitoring Form and guarantee excellent communication at every stage of your form preparation process with airSlate SignNow.
Create this form in 5 minutes or less
Create this form in 5 minutes!
How to create an eSignature for the clinical monitoring form
How to create an electronic signature for a PDF online
How to create an electronic signature for a PDF in Google Chrome
How to create an e-signature for signing PDFs in Gmail
How to create an e-signature right from your smartphone
How to create an e-signature for a PDF on iOS
How to create an e-signature for a PDF on Android
People also ask
-
What is a clinical monitoring form and why is it important?
A clinical monitoring form is a crucial document used in clinical trials to collect and standardize data. It ensures that all necessary information is gathered consistently, which is vital for maintaining the integrity and effectiveness of the trial. Using airSlate SignNow can streamline the creation and management of these forms, providing an efficient way to capture essential clinical data.
-
How does airSlate SignNow enhance the use of clinical monitoring forms?
airSlate SignNow simplifies the process of sending, signing, and managing clinical monitoring forms electronically. With features like easy document sharing and eSignature capabilities, it improves compliance and reduces the risk of errors during data collection. This ensures a smoother workflow for clinical trials and enhances overall efficiency.
-
Are there any costs associated with using airSlate SignNow for clinical monitoring forms?
Yes, there are pricing plans tailored to various business needs, making airSlate SignNow a cost-effective solution for managing clinical monitoring forms. The pricing is designed to accommodate everything from small teams to large organizations. For specific pricing details, it is best to visit the airSlate website or contact their sales team.
-
Can I integrate airSlate SignNow with other software used for clinical trials?
Absolutely! airSlate SignNow offers integrations with various CRM and document management systems, allowing you to streamline the workflow of clinical monitoring forms. This seamless integration helps maintain data consistency across different platforms and enhances the overall efficiency of your clinical trial processes.
-
What are the benefits of using electronic clinical monitoring forms?
Using electronic clinical monitoring forms via airSlate SignNow offers numerous benefits, such as improved accuracy, faster processing, and enhanced compliance with regulatory standards. Digital forms eliminate the need for paper, reducing environmental impact and operational costs. Additionally, electronic signatures remove delays often associated with physical document handling.
-
How secure is the data collected through clinical monitoring forms in airSlate SignNow?
Security is a top priority at airSlate SignNow. The platform uses advanced encryption and secure electronic signature technology to protect sensitive data collected through clinical monitoring forms. Moreover, airSlate is compliant with industry standards, ensuring that your data remains private and secure throughout its lifecycle.
-
Is it easy to customize clinical monitoring forms in airSlate SignNow?
Yes, customizing clinical monitoring forms in airSlate SignNow is user-friendly and straightforward. You can easily create tailored forms that meet your specific clinical trial requirements with a drag-and-drop interface. This flexibility allows you to adapt your forms quickly as your monitoring needs evolve.
Get more for Clinical Monitoring Form
- Parenthetical title example form
- Box whisker worksheet flyparsonsorg form
- Request for elm 650 mediation form
- Field trip medication form humble independent school district
- Field trip permission form salembgc org salembgc
- United states bureau of foreign and domestic commercethe form
- Fhcs housing counseling program disclosure form generic image fairhousingakron
- 2092 contingency for sale and closing of buyers property v10 04 sampleqxp contact attorneys deskbook form
Find out other Clinical Monitoring Form
- How To Electronic signature Alabama Business Operations Form
- Help Me With Electronic signature Alabama Car Dealer Presentation
- How Can I Electronic signature California Car Dealer PDF
- How Can I Electronic signature California Car Dealer Document
- How Can I Electronic signature Colorado Car Dealer Form
- How To Electronic signature Florida Car Dealer Word
- How Do I Electronic signature Florida Car Dealer Document
- Help Me With Electronic signature Florida Car Dealer Presentation
- Can I Electronic signature Georgia Car Dealer PDF
- How Do I Electronic signature Georgia Car Dealer Document
- Can I Electronic signature Georgia Car Dealer Form
- Can I Electronic signature Idaho Car Dealer Document
- How Can I Electronic signature Illinois Car Dealer Document
- How Can I Electronic signature North Carolina Banking PPT
- Can I Electronic signature Kentucky Car Dealer Document
- Can I Electronic signature Louisiana Car Dealer Form
- How Do I Electronic signature Oklahoma Banking Document
- How To Electronic signature Oklahoma Banking Word
- How Can I Electronic signature Massachusetts Car Dealer PDF
- How Can I Electronic signature Michigan Car Dealer Document