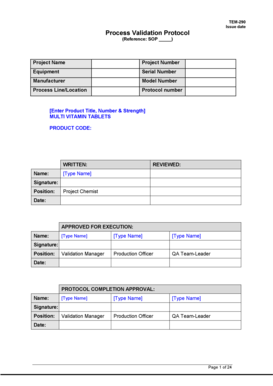
Validation Protocol Template Form


What is the HVAC Validation Protocol Template
The HVAC validation protocol template is a structured document designed to ensure that heating, ventilation, and air conditioning systems meet specific performance standards and regulatory requirements. This template serves as a guideline for conducting validation activities, documenting results, and ensuring compliance with industry standards. It outlines the necessary steps to verify that HVAC systems operate effectively and safely, contributing to overall building performance and occupant comfort.
Key Elements of the HVAC Validation Protocol Template
Essential components of the HVAC validation protocol template include:
- Purpose and Scope: Clearly defines the objectives of the validation process and the systems involved.
- Validation Plan: Outlines the methodology for conducting validation, including testing procedures and criteria for success.
- Documentation Requirements: Specifies the necessary records to maintain throughout the validation process, ensuring traceability and accountability.
- Acceptance Criteria: Defines the benchmarks that must be met for the HVAC system to be considered validated.
- Roles and Responsibilities: Identifies the personnel involved in the validation process and their specific duties.
Steps to Complete the HVAC Validation Protocol Template
Completing the HVAC validation protocol template involves several key steps:
- Define Objectives: Clearly articulate the goals of the validation process.
- Gather Documentation: Collect all relevant documents, including system specifications and previous validation reports.
- Conduct Testing: Perform the necessary tests as outlined in the validation plan to assess system performance.
- Analyze Results: Review the data collected during testing to determine if the system meets acceptance criteria.
- Document Findings: Record all results and observations in the template, ensuring thorough documentation of the validation process.
- Review and Approve: Have the completed protocol reviewed by relevant stakeholders for approval.
Legal Use of the HVAC Validation Protocol Template
Utilizing the HVAC validation protocol template legally requires adherence to applicable regulations and standards governing HVAC systems. Compliance with local, state, and federal guidelines is essential to ensure that the validation process is recognized as valid. This includes following industry best practices and maintaining accurate records that can be referenced in case of audits or inspections.
How to Use the HVAC Validation Protocol Template
To effectively use the HVAC validation protocol template, follow these guidelines:
- Customize the Template: Modify the template to fit the specific requirements of your HVAC system and organizational standards.
- Involve Stakeholders: Engage all relevant parties in the validation process to ensure comprehensive input and adherence to standards.
- Regularly Update: Keep the template current by revising it to reflect changes in regulations, technology, or organizational policies.
Examples of Using the HVAC Validation Protocol Template
Examples of the HVAC validation protocol template in action include:
- New Installations: Validating a newly installed HVAC system to ensure it operates according to specifications.
- System Upgrades: Documenting the validation of an upgraded system to verify that enhancements meet performance standards.
- Routine Maintenance: Using the template to validate HVAC systems during scheduled maintenance to ensure ongoing compliance and performance.
Quick guide on how to complete validation protocol
Complete validation protocol effortlessly on any device
Online document administration has gained popularity among businesses and individuals. It offers a perfect environmentally friendly alternative to conventional printed and signed documents, as you can easily access the appropriate form and securely store it online. airSlate SignNow equips you with all the resources necessary to create, edit, and eSign your documents swiftly without interruptions. Manage hvac validation protocol template on any platform with airSlate SignNow Android or iOS applications and streamline any document-related procedure today.
How to edit and eSign hvac performance qualification protocol template with ease
- Obtain analytical method validation protocol template and then click Get Form to begin.
- Employ the tools we provide to finalize your document.
- Highlight pertinent sections of your documents or obscure sensitive details with tools that airSlate SignNow supplies specifically for that purpose.
- Create your eSignature using the Sign feature, which takes mere seconds and holds the same legal validity as a traditional handwritten signature.
- Review the information and then click the Done button to save your modifications.
- Choose how you wish to send your form, via email, text message (SMS), or invitation link, or download it to your computer.
Put an end to lost or mislaid documents, the tedious task of searching for forms, or mistakes that require printing new copies. airSlate SignNow meets all your document management requirements in just a few clicks from any device you prefer. Edit and eSign process validation protocol and ensure exceptional communication at every stage of your form preparation process with airSlate SignNow.
Create this form in 5 minutes or less
Create this form in 5 minutes!
How to create an eSignature for the process validation template
How to create an electronic signature for a PDF online
How to create an electronic signature for a PDF in Google Chrome
How to create an e-signature for signing PDFs in Gmail
How to create an e-signature right from your smartphone
How to create an e-signature for a PDF on iOS
How to create an e-signature for a PDF on Android
People also ask hvac validation protocol template
-
What is an HVAC validation protocol template?
An HVAC validation protocol template is a structured document designed to ensure compliance and quality assurance for HVAC systems. It outlines the necessary steps for validating performance, verifying specifications, and maintaining operational standards in various environments.
-
How can the HVAC validation protocol template benefit my business?
Using an HVAC validation protocol template can streamline your validation processes, ensuring that your HVAC systems operate efficiently and comply with industry regulations. This not only enhances system reliability but also can lead to cost savings by preventing failures and reducing downtime.
-
Is the HVAC validation protocol template customizable?
Yes, the HVAC validation protocol template is customizable to meet your specific business needs. You can modify sections to include particular parameters, standards, and procedures relevant to your operational requirements, ensuring that it accurately reflects your processes.
-
What features does airSlate SignNow offer for the HVAC validation protocol template?
AirSlate SignNow provides features such as easy document creation, eSignature integration, and document tracking for your HVAC validation protocol template. These functionalities allow for seamless collaboration and ensure all stakeholders can access and sign documents efficiently.
-
Can I integrate the HVAC validation protocol template with other software?
Absolutely! The HVAC validation protocol template can be integrated with a range of software solutions, including project management and compliance platforms. This integration helps maintain records and streamline workflows for HVAC system validation.
-
What is the pricing structure for using airSlate SignNow with the HVAC validation protocol template?
AirSlate SignNow offers a variety of pricing plans designed to fit different business needs, including plans tailored for those utilizing the HVAC validation protocol template. You can choose a plan based on the volume of documents and specific features required, ensuring cost-effectiveness.
-
How does an HVAC validation protocol template help with compliance?
An HVAC validation protocol template ensures adherence to industry standards and regulatory requirements, helping you maintain compliance. By following a structured validation process outlined in the template, you can systematically document and demonstrate compliance during audits.
Get more for hvac performance qualification protocol template
Find out other analytical method validation protocol template
- How Can I eSign North Carolina Non-Profit Document
- How To eSign Vermont Non-Profit Presentation
- How Do I eSign Hawaii Orthodontists PDF
- How Can I eSign Colorado Plumbing PDF
- Can I eSign Hawaii Plumbing PDF
- How Do I eSign Hawaii Plumbing Form
- Can I eSign Hawaii Plumbing Form
- How To eSign Hawaii Plumbing Word
- Help Me With eSign Hawaii Plumbing Document
- How To eSign Hawaii Plumbing Presentation
- How To eSign Maryland Plumbing Document
- How Do I eSign Mississippi Plumbing Word
- Can I eSign New Jersey Plumbing Form
- How Can I eSign Wisconsin Plumbing PPT
- Can I eSign Colorado Real Estate Form
- How To eSign Florida Real Estate Form
- Can I eSign Hawaii Real Estate Word
- How Do I eSign Hawaii Real Estate Word
- How To eSign Hawaii Real Estate Document
- How Do I eSign Hawaii Real Estate Presentation