Mdsap Audit Checklist Excel Form


What is the Mdsap Audit Checklist Excel
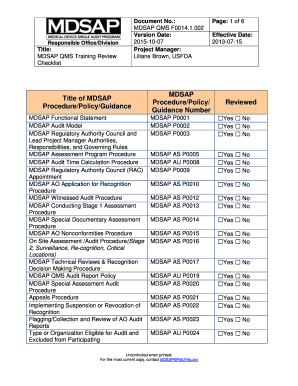
The Mdsap Audit Checklist Excel is a structured tool designed for organizations participating in the Medical Device Single Audit Program (MDSAP). This checklist serves as a comprehensive guide to ensure compliance with international regulatory requirements for medical devices. It includes various criteria that organizations must meet to demonstrate adherence to quality management systems and regulatory standards across multiple jurisdictions, including the United States, Canada, Brazil, Australia, and Japan.
Key elements of the Mdsap Audit Checklist Excel
Understanding the key elements of the Mdsap Audit Checklist Excel is crucial for successful compliance. The checklist typically includes:
- Document Control: Ensures that all documents are properly managed and accessible.
- Risk Management: Involves identifying, evaluating, and mitigating risks associated with medical devices.
- Design and Development: Covers the processes for designing and developing medical devices, ensuring they meet safety and effectiveness standards.
- Production and Process Controls: Focuses on maintaining quality during manufacturing processes.
- Post-Market Surveillance: Involves monitoring the performance of medical devices after they have been released to the market.
Steps to complete the Mdsap Audit Checklist Excel
Completing the Mdsap Audit Checklist Excel involves a systematic approach to ensure thoroughness and accuracy. The steps typically include:
- Gather relevant documentation and resources related to your quality management system.
- Review each section of the checklist, ensuring all criteria are addressed.
- Document evidence of compliance for each requirement, including policies, procedures, and records.
- Engage relevant team members to verify the accuracy of the information provided.
- Conduct a final review to ensure completeness before submission.
How to use the Mdsap Audit Checklist Excel
Using the Mdsap Audit Checklist Excel effectively requires familiarity with its structure and purpose. Begin by opening the Excel file and navigating through the various tabs that represent different sections of the audit. Each section will contain specific requirements and prompts to guide you in providing necessary information. Utilize the built-in features of Excel, such as comments and notes, to clarify points or add additional context as needed. Regularly save your progress to avoid data loss.
Legal use of the Mdsap Audit Checklist Excel
The legal use of the Mdsap Audit Checklist Excel hinges on its compliance with regulatory standards set forth by various health authorities. Organizations must ensure that the checklist is used in accordance with the guidelines established by the MDSAP program. This includes maintaining accurate records, adhering to confidentiality agreements, and ensuring that all information is truthful and complete. Failure to comply with these legal standards may result in penalties or regulatory action.
Examples of using the Mdsap Audit Checklist Excel
Examples of using the Mdsap Audit Checklist Excel can provide practical insights into its application. For instance, a medical device manufacturer may use the checklist to prepare for an upcoming audit by systematically reviewing their quality management practices. Another example could involve a company conducting internal audits to identify areas for improvement before the official MDSAP audit. These examples illustrate how the checklist can facilitate compliance and enhance overall quality management processes.
Quick guide on how to complete mdsap audit checklist excel
Effortlessly Complete Mdsap Audit Checklist Excel on Any Device
Managing documents online has gained popularity among businesses and individuals alike. It offers an ideal eco-friendly alternative to traditional printed and signed forms, allowing you to acquire the needed template and securely save it online. airSlate SignNow equips you with all the tools required to create, modify, and electronically sign your documents swiftly without delays. Handle Mdsap Audit Checklist Excel on any device using the airSlate SignNow Android or iOS applications and enhance any document-driven process today.
How to Modify and eSign Mdsap Audit Checklist Excel with Ease
- Find Mdsap Audit Checklist Excel and select Get Form to begin.
- Utilize the tools we offer to fill out your document.
- Emphasize pertinent sections of your documents or conceal sensitive information with tools that airSlate SignNow provides specifically for that purpose.
- Generate your eSignature using the Sign tool, which takes seconds and carries the same legal validity as a conventional wet ink signature.
- Review the details and click on the Done button to finalize your changes.
- Choose how you wish to send your form, via email, SMS, or invitation link, or download it to your computer.
Eliminate the hassle of lost or misplaced files, tedious document searching, or mistakes that necessitate printing new copies. airSlate SignNow meets your document management needs in just a few clicks from any chosen device. Modify and eSign Mdsap Audit Checklist Excel and ensure excellent communication throughout your document preparation process with airSlate SignNow.
Create this form in 5 minutes or less
Create this form in 5 minutes!
How to create an eSignature for the mdsap audit checklist excel
How to create an electronic signature for a PDF online
How to create an electronic signature for a PDF in Google Chrome
How to create an e-signature for signing PDFs in Gmail
How to create an e-signature right from your smartphone
How to create an e-signature for a PDF on iOS
How to create an e-signature for a PDF on Android
People also ask
-
What is the MDSAP requirements checklist?
The MDSAP requirements checklist is a comprehensive guide that outlines the essential criteria and standards that manufacturers must meet to successfully navigate the Medical Device Single Audit Program. Utilizing this checklist can streamline your compliance process and ensure that your documentation aligns with the necessary regulations.
-
How can airSlate SignNow help with the MDSAP requirements checklist?
airSlate SignNow provides an efficient platform to manage and store crucial documents required for the MDSAP requirements checklist. With its robust eSigning and document management capabilities, your team can easily collaborate, track compliance, and maintain thorough records without the hassle of traditional paperwork.
-
Is there a cost associated with using airSlate SignNow for MDSAP compliance?
Yes, airSlate SignNow offers several pricing plans to suit different business needs, and utilizing it to address MDSAP requirements can be a cost-effective solution. By automating document workflows associated with the MDSAP requirements checklist, your organization can save both time and resources in the long run.
-
What features of airSlate SignNow support MDSAP compliance?
airSlate SignNow includes features such as secure eSignature, customizable templates, and document tracking that can signNowly aid in meeting MDSAP requirements checklist. These features facilitate efficient document management, ensuring that all compliance-related materials are easily accessible and properly handled.
-
Can I integrate airSlate SignNow with other tools for MDSAP documentation?
Absolutely! airSlate SignNow offers integrations with a variety of third-party applications that can enhance your compliance efforts around the MDSAP requirements checklist. Whether you use project management tools or CRM systems, these integrations can help streamline your document workflow further.
-
How does airSlate SignNow ensure document security for MDSAP compliance?
With airSlate SignNow, document security is a top priority. The platform employs advanced encryption, secure cloud storage, and compliance with industry standards to protect sensitive documents while addressing MDSAP requirements checklist. You can be confident that your compliance documents are safe and secure at all times.
-
What are the benefits of using airSlate SignNow for MDSAP compliance?
Using airSlate SignNow for MDSAP compliance offers numerous benefits, including increased efficiency, reduced turnaround times for document approval, and improved accuracy in documentation. By leveraging this platform, businesses can more easily adhere to the MDSAP requirements checklist and focus on their core operations.
Get more for Mdsap Audit Checklist Excel
- Cc 1477 writ of fieri facias form
- Metropolitan nashville police department authorization to enforce form
- Criminal trespass waiver mnpd form 188 fillable
- Same time you file for your minor child you should use the civil division packet 10551 how form
- How to ask the court to change a name in the chancery division family part how to ask the court to change a name in the form
- Infant daily production record meal documentation facility form
- Dwc form 153
- Filing a complaint to start your case civil law self help centersample complaint letter templateusagovfile an online form
Find out other Mdsap Audit Checklist Excel
- Electronic signature Finance & Tax Accounting Word South Carolina Later
- How Do I Electronic signature Illinois Healthcare / Medical Purchase Order Template
- Electronic signature Louisiana Healthcare / Medical Quitclaim Deed Online
- Electronic signature Louisiana Healthcare / Medical Quitclaim Deed Computer
- How Do I Electronic signature Louisiana Healthcare / Medical Limited Power Of Attorney
- Electronic signature Maine Healthcare / Medical Letter Of Intent Fast
- How To Electronic signature Mississippi Healthcare / Medical Month To Month Lease
- Electronic signature Nebraska Healthcare / Medical RFP Secure
- Electronic signature Nevada Healthcare / Medical Emergency Contact Form Later
- Electronic signature New Hampshire Healthcare / Medical Credit Memo Easy
- Electronic signature New Hampshire Healthcare / Medical Lease Agreement Form Free
- Electronic signature North Dakota Healthcare / Medical Notice To Quit Secure
- Help Me With Electronic signature Ohio Healthcare / Medical Moving Checklist
- Electronic signature Education PPT Ohio Secure
- Electronic signature Tennessee Healthcare / Medical NDA Now
- Electronic signature Tennessee Healthcare / Medical Lease Termination Letter Online
- Electronic signature Oklahoma Education LLC Operating Agreement Fast
- How To Electronic signature Virginia Healthcare / Medical Contract
- How To Electronic signature Virginia Healthcare / Medical Operating Agreement
- Electronic signature Wisconsin Healthcare / Medical Business Letter Template Mobile