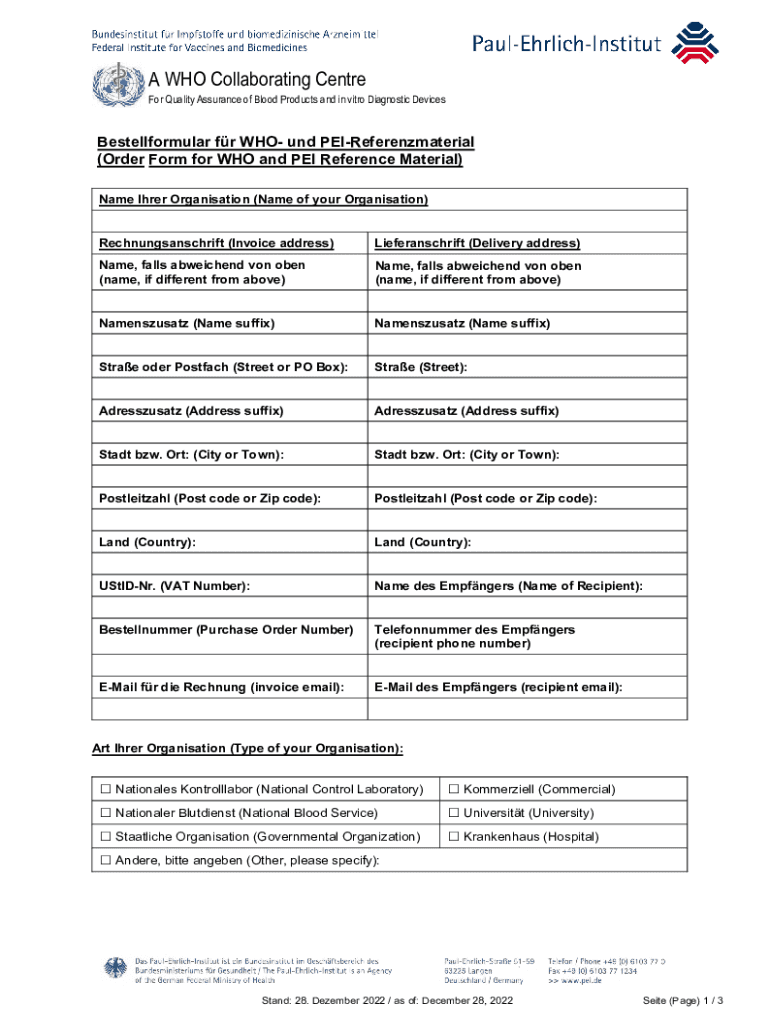
Guide to the Preparation, Use and Quality Assurance of Form


Understanding the Guide to the Preparation, Use and Quality Assurance
The Guide to the Preparation, Use and Quality Assurance provides essential information for individuals and businesses looking to navigate the complexities of document preparation and quality assurance processes. This guide outlines the necessary steps and considerations to ensure that documents are prepared accurately and meet quality standards. It serves as a comprehensive resource for understanding the requirements and best practices associated with various forms and documents.
Steps to Effectively Use the Guide
To effectively utilize the Guide to the Preparation, Use and Quality Assurance, follow these steps:
- Review the guide thoroughly to understand its structure and key components.
- Identify the specific documents or forms relevant to your needs.
- Follow the outlined procedures for preparation and submission.
- Ensure compliance with quality assurance measures detailed in the guide.
- Keep abreast of any updates or changes to the guidelines that may affect your documentation process.
Key Elements of the Guide
Several key elements are integral to the Guide to the Preparation, Use and Quality Assurance:
- Document preparation standards that outline the necessary information and format.
- Quality assurance protocols to verify the accuracy and completeness of documents.
- Legal considerations that ensure compliance with applicable laws and regulations.
- Submission methods, detailing how to file documents online, by mail, or in person.
Legal Use of the Guide
The Guide to the Preparation, Use and Quality Assurance is designed to comply with legal standards in the United States. It provides users with the necessary information to ensure that their documents meet federal and state requirements. Understanding the legal implications of document preparation is crucial, as non-compliance can lead to penalties or legal disputes.
Required Documents for Compliance
When using the Guide to the Preparation, Use and Quality Assurance, it is important to gather all required documents. These may include:
- Identification documents, such as a driver's license or passport.
- Financial records relevant to the form being completed.
- Supporting documentation that substantiates claims made in the forms.
Ensuring that all required documents are in order will facilitate a smoother preparation and submission process.
Examples of Application in Real Scenarios
Real-world examples can illustrate how the Guide to the Preparation, Use and Quality Assurance is applied:
- A small business preparing tax documents using the guide to ensure compliance with IRS regulations.
- An individual applying for a government grant, utilizing the guide to prepare necessary forms accurately.
- A nonprofit organization ensuring that its documentation meets quality assurance standards to maintain funding eligibility.
Form Submission Methods
The Guide to the Preparation, Use and Quality Assurance outlines various submission methods for forms, including:
- Online submission through designated platforms.
- Mailing physical copies to the appropriate agencies.
- In-person submission at designated offices or agencies.
Understanding these methods is crucial for ensuring that documents are submitted correctly and on time.
Quick guide on how to complete guide to the preparation use and quality assurance of
Effortlessly Prepare Guide To The Preparation, Use And Quality Assurance Of on Any Device
Managing documents online has become increasingly popular among businesses and individuals alike. It offers an ideal eco-friendly substitute for traditional printed and signed papers, allowing you to access the necessary forms and securely store them online. airSlate SignNow equips you with all the resources required to create, edit, and electronically sign your documents swiftly and without delays. Handle Guide To The Preparation, Use And Quality Assurance Of on any device using the airSlate SignNow apps for Android or iOS and streamline any document-related procedure today.
The easiest way to edit and eSign Guide To The Preparation, Use And Quality Assurance Of seamlessly
- Find Guide To The Preparation, Use And Quality Assurance Of and click Get Form to begin.
- Utilize the tools we provide to fill out your document.
- Emphasize pertinent sections of the documents or obscure sensitive data using the tools that airSlate SignNow specifically offers for that purpose.
- Create your eSignature with the Sign tool, which only takes seconds and carries the same legal validity as a conventional ink signature.
- Review the information and click on the Done button to save your modifications.
- Choose how you wish to send your form—via email, SMS, an invitation link, or download it to your computer.
Stop worrying about lost or misplaced documents, the hassle of searching for forms, or errors that necessitate printing new copies. airSlate SignNow fulfills all your document management needs in just a few clicks from any device of your preference. Modify and eSign Guide To The Preparation, Use And Quality Assurance Of to ensure excellent communication at every stage of the form preparation process with airSlate SignNow.
Create this form in 5 minutes or less
Create this form in 5 minutes!
How to create an eSignature for the guide to the preparation use and quality assurance of
How to create an electronic signature for a PDF online
How to create an electronic signature for a PDF in Google Chrome
How to create an e-signature for signing PDFs in Gmail
How to create an e-signature right from your smartphone
How to create an e-signature for a PDF on iOS
How to create an e-signature for a PDF on Android
Get more for Guide To The Preparation, Use And Quality Assurance Of
- Form 4822 1651193
- Unit 2 test download ebook and pdf files babushkabooks english land form
- Quantum commander form
- Examples of ada medical release forms
- Rha rental application form
- Mto medical form
- Pennsylvania income tax return pa 40 department of form
- The ben and catherine ivy center for advanced brain tumor form
Find out other Guide To The Preparation, Use And Quality Assurance Of
- How To Electronic signature Arkansas Real Estate Contract
- Electronic signature Idaho Plumbing Claim Myself
- Electronic signature Kansas Plumbing Business Plan Template Secure
- Electronic signature Louisiana Plumbing Purchase Order Template Simple
- Can I Electronic signature Wyoming Legal Limited Power Of Attorney
- How Do I Electronic signature Wyoming Legal POA
- How To Electronic signature Florida Real Estate Contract
- Electronic signature Florida Real Estate NDA Secure
- Can I Electronic signature Florida Real Estate Cease And Desist Letter
- How Can I Electronic signature Hawaii Real Estate LLC Operating Agreement
- Electronic signature Georgia Real Estate Letter Of Intent Myself
- Can I Electronic signature Nevada Plumbing Agreement
- Electronic signature Illinois Real Estate Affidavit Of Heirship Easy
- How To Electronic signature Indiana Real Estate Quitclaim Deed
- Electronic signature North Carolina Plumbing Business Letter Template Easy
- Electronic signature Kansas Real Estate Residential Lease Agreement Simple
- How Can I Electronic signature North Carolina Plumbing Promissory Note Template
- Electronic signature North Dakota Plumbing Emergency Contact Form Mobile
- Electronic signature North Dakota Plumbing Emergency Contact Form Easy
- Electronic signature Rhode Island Plumbing Business Plan Template Later