CG 5353 MONTHLY REPORT for NARCOTICS and OTHER CONTROLLED DRUGS 2020-2026


What is the CG 5353 Monthly Report for Narcotics and Other Controlled Drugs
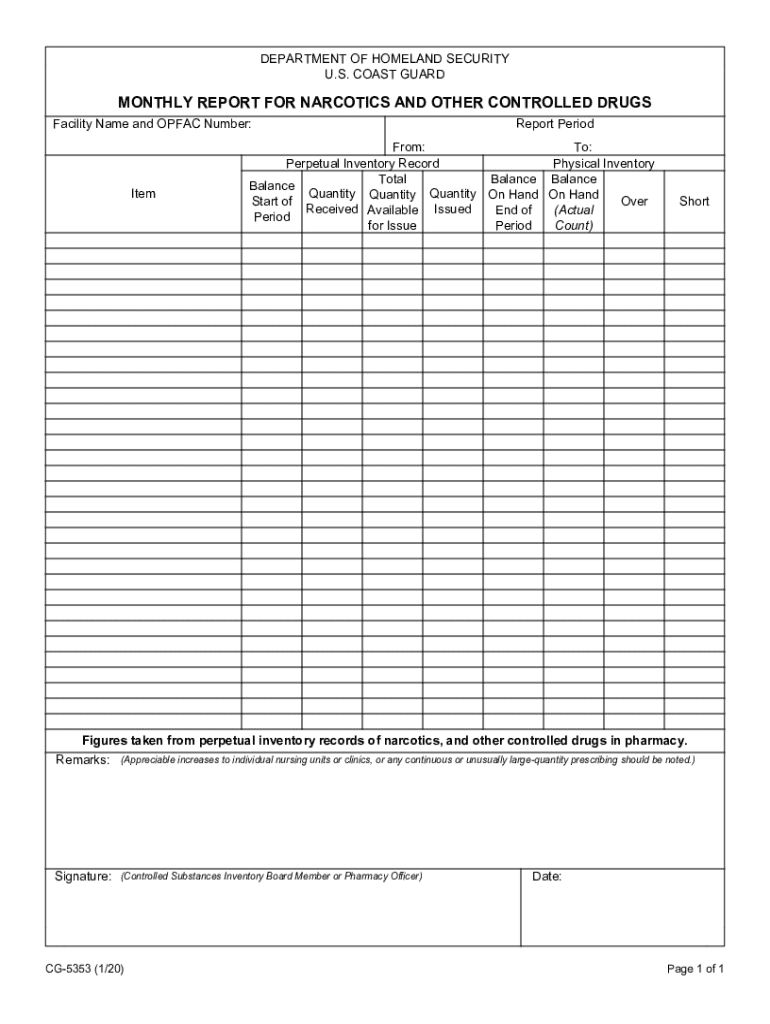
The CG 5353 is a critical form used for reporting the monthly inventory and transactions involving narcotics and other controlled substances. This report is essential for healthcare providers and facilities that handle these drugs, ensuring compliance with federal regulations. The form tracks the receipt, distribution, and disposal of controlled substances, providing a comprehensive overview of a facility's narcotics management.
How to Use the CG 5353 Monthly Report for Narcotics and Other Controlled Drugs
Using the CG 5353 involves several steps that ensure accurate reporting. First, gather all necessary data regarding narcotics handled during the month, including quantities received, dispensed, and disposed of. Next, complete the form by entering this information in the designated sections. It is crucial to maintain accurate records to support the data reported on the CG 5353, as this information may be subject to audits by regulatory agencies.
Steps to Complete the CG 5353 Monthly Report for Narcotics and Other Controlled Drugs
Completing the CG 5353 requires careful attention to detail. Follow these steps:
- Collect all relevant data regarding narcotics transactions for the month.
- Fill in the form with accurate quantities of drugs received, dispensed, and disposed of.
- Ensure that all entries are legible and correctly formatted.
- Review the completed form for any errors or omissions.
- Submit the form by the required deadline to the appropriate regulatory body.
Legal Use of the CG 5353 Monthly Report for Narcotics and Other Controlled Drugs
The CG 5353 must be used in accordance with federal and state regulations governing controlled substances. Healthcare providers are legally obligated to maintain accurate records of narcotics transactions and report them monthly. Failure to comply with these regulations can result in penalties, including fines and potential loss of licensure. It is essential to understand the legal implications of the information reported on this form.
Key Elements of the CG 5353 Monthly Report for Narcotics and Other Controlled Drugs
Key elements of the CG 5353 include:
- Facility information, including name and address.
- Details of controlled substances handled, including drug names and quantities.
- Sections for documenting receipts, distributions, and disposals.
- Signature of the responsible individual verifying the accuracy of the report.
Form Submission Methods for the CG 5353 Monthly Report for Narcotics and Other Controlled Drugs
The CG 5353 can be submitted through various methods, including:
- Online submission via designated government portals.
- Mailing a physical copy to the appropriate regulatory agency.
- In-person submission at local regulatory offices.
Quick guide on how to complete cg 5353 monthly report for narcotics and other controlled drugs
Prepare CG 5353 MONTHLY REPORT FOR NARCOTICS AND OTHER CONTROLLED DRUGS effortlessly on any device
Online document management has gained popularity among businesses and individuals. It offers an ideal eco-friendly alternative to conventional printed and signed documents, as you can easily find the correct form and securely store it online. airSlate SignNow equips you with all the tools necessary to create, edit, and eSign your documents promptly without delays. Manage CG 5353 MONTHLY REPORT FOR NARCOTICS AND OTHER CONTROLLED DRUGS on any platform using airSlate SignNow’s Android or iOS applications and enhance any document-centric operation today.
The easiest way to modify and eSign CG 5353 MONTHLY REPORT FOR NARCOTICS AND OTHER CONTROLLED DRUGS seamlessly
- Find CG 5353 MONTHLY REPORT FOR NARCOTICS AND OTHER CONTROLLED DRUGS and click on Get Form to begin.
- Utilize the tools we provide to complete your form.
- Highlight pertinent sections of your documents or blackout sensitive information with tools specifically designed by airSlate SignNow for that purpose.
- Generate your eSignature with the Sign feature, which takes seconds and holds the same legal validity as a traditional handwritten signature.
- Review the information and click on the Done button to save your changes.
- Choose your preferred method to send your form: via email, SMS, invitation link, or download it to your computer.
Say goodbye to lost or misplaced documents, tiring form searches, or errors that require printing new copies. airSlate SignNow takes care of all your document management needs with just a few clicks from any device you prefer. Edit and eSign CG 5353 MONTHLY REPORT FOR NARCOTICS AND OTHER CONTROLLED DRUGS and ensure excellent communication at every stage of the form preparation process with airSlate SignNow.
Create this form in 5 minutes or less
Find and fill out the correct cg 5353 monthly report for narcotics and other controlled drugs
Create this form in 5 minutes!
How to create an eSignature for the cg 5353 monthly report for narcotics and other controlled drugs
How to create an electronic signature for a PDF online
How to create an electronic signature for a PDF in Google Chrome
How to create an e-signature for signing PDFs in Gmail
How to create an e-signature right from your smartphone
How to create an e-signature for a PDF on iOS
How to create an e-signature for a PDF on Android
People also ask
-
What is cg 5353 and how does it relate to airSlate SignNow?
The cg 5353 is a specific feature within airSlate SignNow that enhances document management and eSigning capabilities. It allows users to streamline their workflows and ensure compliance with industry standards. By utilizing cg 5353, businesses can improve efficiency and reduce turnaround times for document processing.
-
How much does airSlate SignNow cost with cg 5353 features?
Pricing for airSlate SignNow varies based on the plan selected, but all plans include access to the cg 5353 features. Businesses can choose from different tiers that cater to their specific needs, ensuring they get the best value for their investment in document management solutions.
-
What are the key benefits of using cg 5353 in airSlate SignNow?
The cg 5353 feature in airSlate SignNow offers numerous benefits, including enhanced security, improved user experience, and faster document turnaround times. By leveraging cg 5353, businesses can ensure that their documents are signed and processed efficiently, leading to increased productivity and customer satisfaction.
-
Can I integrate cg 5353 with other software applications?
Yes, airSlate SignNow, including the cg 5353 feature, offers seamless integrations with various software applications. This allows businesses to connect their existing tools and workflows, enhancing overall efficiency and ensuring that document management fits smoothly into their operations.
-
Is cg 5353 suitable for small businesses?
Absolutely! The cg 5353 feature in airSlate SignNow is designed to be user-friendly and cost-effective, making it ideal for small businesses. It provides essential eSigning and document management capabilities without the complexity or high costs associated with other solutions.
-
What types of documents can I manage with cg 5353?
With cg 5353 in airSlate SignNow, you can manage a wide variety of documents, including contracts, agreements, and forms. This versatility allows businesses to handle all their document needs in one place, ensuring a streamlined process from creation to signing.
-
How does cg 5353 enhance security for my documents?
The cg 5353 feature in airSlate SignNow includes advanced security measures such as encryption and secure access controls. These features help protect sensitive information and ensure that only authorized users can access and sign documents, providing peace of mind for businesses.
Get more for CG 5353 MONTHLY REPORT FOR NARCOTICS AND OTHER CONTROLLED DRUGS
Find out other CG 5353 MONTHLY REPORT FOR NARCOTICS AND OTHER CONTROLLED DRUGS
- eSign Hawaii Plumbing Letter Of Intent Myself
- eSign Hawaii Plumbing Letter Of Intent Fast
- Help Me With eSign Idaho Plumbing Profit And Loss Statement
- eSign Illinois Plumbing Letter Of Intent Now
- eSign Massachusetts Orthodontists Last Will And Testament Now
- eSign Illinois Plumbing Permission Slip Free
- eSign Kansas Plumbing LLC Operating Agreement Secure
- eSign Kentucky Plumbing Quitclaim Deed Free
- eSign Legal Word West Virginia Online
- Can I eSign Wisconsin Legal Warranty Deed
- eSign New Hampshire Orthodontists Medical History Online
- eSign Massachusetts Plumbing Job Offer Mobile
- How To eSign Pennsylvania Orthodontists Letter Of Intent
- eSign Rhode Island Orthodontists Last Will And Testament Secure
- eSign Nevada Plumbing Business Letter Template Later
- eSign Nevada Plumbing Lease Agreement Form Myself
- eSign Plumbing PPT New Jersey Later
- eSign New York Plumbing Rental Lease Agreement Simple
- eSign North Dakota Plumbing Emergency Contact Form Mobile
- How To eSign North Dakota Plumbing Emergency Contact Form