Glucometer Quality Control Log Template Form


What is the glucometer quality control log template
The glucometer quality control log template is a structured document designed to track the accuracy and performance of glucometers used in diabetes management. This template allows healthcare providers and patients to record the calibration results and quality control checks performed on glucometers. It ensures that the devices are functioning correctly, which is essential for reliable blood glucose readings. By maintaining a detailed log, users can identify trends, troubleshoot issues, and ensure compliance with health regulations.
How to use the glucometer quality control log template
Using the glucometer quality control log template involves several straightforward steps. First, gather all necessary information, including the date, time, and type of control solution used. Next, perform the quality control test as per the glucometer's instructions and record the results in the template. It is crucial to include any observations or anomalies during the testing process. Regularly updating the log after each test helps maintain an accurate history of the glucometer's performance, which is vital for effective diabetes management.
Key elements of the glucometer quality control log template
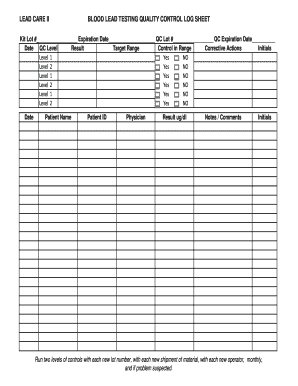
The key elements of the glucometer quality control log template typically include the following:
- Date and Time: When the quality control test was conducted.
- Control Solution Lot Number: Identifies the batch of control solution used.
- Test Results: The numerical results obtained from the quality control test.
- Operator's Name: The individual who performed the test.
- Comments: Any additional notes regarding the test or device performance.
These elements are essential for maintaining accurate records and ensuring compliance with health standards.
Steps to complete the glucometer quality control log template
Completing the glucometer quality control log template involves a series of methodical steps:
- Gather the necessary control solutions and equipment.
- Perform the quality control test as directed by the glucometer's manufacturer.
- Record the date, time, and results of the test in the template.
- Include any relevant comments or observations about the testing process.
- Store the completed log securely for future reference and compliance checks.
Following these steps ensures that the log is comprehensive and useful for ongoing diabetes management.
Legal use of the glucometer quality control log template
The glucometer quality control log template serves a critical role in legal compliance within healthcare settings. It provides documented evidence that quality control measures are being performed regularly, which can be essential during audits or inspections. Maintaining accurate logs helps demonstrate adherence to regulatory standards set forth by organizations such as the Food and Drug Administration (FDA) and the Centers for Medicare & Medicaid Services (CMS). Proper documentation can protect healthcare providers from liability and ensure that patients receive safe and effective care.
Examples of using the glucometer quality control log template
Examples of using the glucometer quality control log template can vary based on the setting. In a clinical environment, healthcare professionals may use the log to document daily quality control tests for multiple glucometers. In a home setting, individuals managing diabetes may use the log to track their glucometer's performance over time, noting any discrepancies in readings. Both scenarios highlight the importance of consistent logging for accurate diabetes management and compliance with health regulations.
Quick guide on how to complete glucometer quality control log template
Prepare Glucometer Quality Control Log Template seamlessly on any device
Online document management has become increasingly prevalent among businesses and individuals. It offers an ideal eco-friendly alternative to conventional printed and signed documents, as you can easily locate the correct form and securely store it online. airSlate SignNow equips you with all the tools necessary to create, alter, and eSign your documents swiftly without delays. Manage Glucometer Quality Control Log Template on any platform with the airSlate SignNow Android or iOS applications and enhance any document-related process today.
The easiest way to modify and eSign Glucometer Quality Control Log Template effortlessly
- Locate Glucometer Quality Control Log Template and click Get Form to begin.
- Utilize the tools we provide to complete your form.
- Emphasize pertinent sections of your documents or obscure sensitive information with tools that airSlate SignNow offers specifically for that purpose.
- Create your signature using the Sign tool, which takes moments and carries the same legal validity as a traditional wet ink signature.
- Confirm the information and then click the Done button to save your changes.
- Select how you wish to send your form, via email, SMS, or invite link, or download it to your computer.
Say goodbye to lost or forgotten documents, tiresome form searches, or errors that necessitate printing new document copies. airSlate SignNow fulfills all your document management requirements in just a few clicks from any device of your choice. Modify and eSign Glucometer Quality Control Log Template and guarantee effective communication at every stage of your form preparation process with airSlate SignNow.
Create this form in 5 minutes or less
Create this form in 5 minutes!
How to create an eSignature for the glucometer quality control log template
How to create an electronic signature for a PDF online
How to create an electronic signature for a PDF in Google Chrome
How to create an e-signature for signing PDFs in Gmail
How to create an e-signature right from your smartphone
How to create an e-signature for a PDF on iOS
How to create an e-signature for a PDF on Android
People also ask
-
What is a glucometer quality control log template?
A glucometer quality control log template is a structured document designed to help healthcare professionals track and maintain the accuracy of blood glucose meters. It allows users to record control test results and ensure that the glucometer is functioning properly. Using this template can enhance the reliability of diabetes management.
-
How can I create a glucometer quality control log template using airSlate SignNow?
Creating a glucometer quality control log template in airSlate SignNow is simple and efficient. You can start by choosing a pre-designed template and customizing it to meet your specific needs. The intuitive platform allows for easy editing and sharing, making quality control documentation streamlined.
-
What are the benefits of using a glucometer quality control log template?
Using a glucometer quality control log template helps ensure that your glucose monitoring is accurate and reliable. It provides a consistent format for recording control solutions and meter tests, which can lead to better patient outcomes. Additionally, it simplifies compliance with regulatory standards.
-
Is the glucometer quality control log template cost-effective?
Yes, the glucometer quality control log template offered by airSlate SignNow is a cost-effective solution for managing glucose testing. By using our digital platform, you eliminate the need for paper logs and related expenses. This also enhances data accuracy and accessibility at a lower operational cost.
-
Can I integrate the glucometer quality control log template with other software?
Absolutely! The glucometer quality control log template can seamlessly integrate with various healthcare management systems and applications. This integration allows you to centralize your data, improving the efficiency of record-keeping and ensuring that all necessary information is readily available.
-
What features should I look for in a glucometer quality control log template?
When looking for a glucometer quality control log template, consider features such as customization options, ease of sharing, and compliance with healthcare regulations. Additionally, check for integration capabilities with other software and tools, as well as user-friendliness to facilitate tracking and management.
-
How does a glucometer quality control log template improve patient care?
Utilizing a glucometer quality control log template improves patient care by ensuring that glucose readings are accurate and reliable. This leads to better-informed treatment decisions and helps manage diabetes more effectively. Furthermore, consistent documentation can enhance communication with healthcare providers about any potential issues.
Get more for Glucometer Quality Control Log Template
- Revised himat no stair items form
- Name period date stephen f austin state university form
- Rcmp grc 5774 page 1 ofde 3 city of port coquitlam form
- Security guard training certification form tn gov tn
- Evaluation assessment form
- Capitalization rules 1 6 name worksheet 1 part i in the form
- Application for senior membership in the charter number form
- How ampamp where to report va abuse and fraud disability happens form
Find out other Glucometer Quality Control Log Template
- Can I eSignature West Virginia Lawers Cease And Desist Letter
- eSignature Alabama Plumbing Confidentiality Agreement Later
- How Can I eSignature Wyoming Lawers Quitclaim Deed
- eSignature California Plumbing Profit And Loss Statement Easy
- How To eSignature California Plumbing Business Letter Template
- eSignature Kansas Plumbing Lease Agreement Template Myself
- eSignature Louisiana Plumbing Rental Application Secure
- eSignature Maine Plumbing Business Plan Template Simple
- Can I eSignature Massachusetts Plumbing Business Plan Template
- eSignature Mississippi Plumbing Emergency Contact Form Later
- eSignature Plumbing Form Nebraska Free
- How Do I eSignature Alaska Real Estate Last Will And Testament
- Can I eSignature Alaska Real Estate Rental Lease Agreement
- eSignature New Jersey Plumbing Business Plan Template Fast
- Can I eSignature California Real Estate Contract
- eSignature Oklahoma Plumbing Rental Application Secure
- How Can I eSignature Connecticut Real Estate Quitclaim Deed
- eSignature Pennsylvania Plumbing Business Plan Template Safe
- eSignature Florida Real Estate Quitclaim Deed Online
- eSignature Arizona Sports Moving Checklist Now