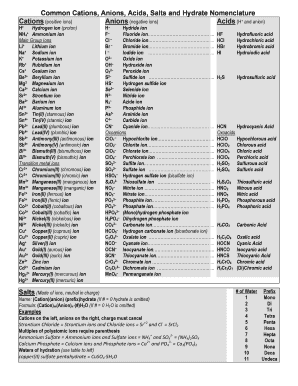
Common Cations, Anions, Acids, Salts and Hydrate Nomenclature Form


Understanding Common Cations and Anions
Cations and anions are essential components in chemistry, representing positively and negatively charged ions, respectively. Common cations include sodium (Na+), potassium (K+), and calcium (Ca2+), while common anions include chloride (Cl-), sulfate (SO42-), and nitrate (NO3-). Understanding these ions is crucial for various applications in science and industry, including the formulation of salts, acids, and hydrates.
How to Use Cation and Anion Nomenclature
Nomenclature for cations and anions follows specific rules that help in identifying and naming these ions correctly. For cations, the name is typically derived from the element name, often with the addition of the word "ion." For example, Na+ is called sodium ion. Anions are named by taking the root of the element name and adding the suffix "-ide" for simple anions, such as chloride for Cl-. For polyatomic ions, specific names are used, such as sulfate for SO42-.
Steps to Complete Cation and Anion Nomenclature
To accurately name cations and anions, follow these steps:
- Identify the element or polyatomic ion.
- Determine if the ion is positively or negatively charged.
- Use the appropriate naming convention based on the ion type.
- For polyatomic ions, refer to a reliable list for their specific names.
- Double-check the charge to ensure accuracy in naming.
Examples of Cation and Anion Nomenclature
Here are some examples to illustrate the nomenclature of common cations and anions:
- Sodium ion (Na+)
- Calcium ion (Ca2+)
- Chloride ion (Cl-)
- Sulfate ion (SO42-)
- Nitrate ion (NO3-)
Legal Use of Cation and Anion Nomenclature
In various scientific and industrial contexts, the accurate use of cation and anion nomenclature is legally significant. For instance, in regulatory documents related to chemical safety, precise naming can impact compliance with environmental laws. Ensuring that the correct nomenclature is used helps avoid legal issues and ensures clarity in communication among professionals in the field.
Key Elements of Cation and Anion Nomenclature
Key elements that define cation and anion nomenclature include:
- Charge of the ion
- Type of ion (monatomic or polyatomic)
- Standard naming conventions
- Consistency in usage across different contexts
Quick guide on how to complete common cations anions acids salts and hydrate nomenclature
Effortlessly Complete Common Cations, Anions, Acids, Salts And Hydrate Nomenclature on Any Device
Digital document management has gained popularity among businesses and individuals. It serves as an ideal eco-friendly substitute for traditional printed and signed documents, allowing you to locate the correct form and securely store it online. airSlate SignNow provides all the resources necessary for you to create, modify, and electronically sign your documents quickly and without delays. Manage Common Cations, Anions, Acids, Salts And Hydrate Nomenclature on any platform with airSlate SignNow's Android or iOS applications and enhance any document-focused operation today.
How to Modify and eSign Common Cations, Anions, Acids, Salts And Hydrate Nomenclature Effortlessly
- Locate Common Cations, Anions, Acids, Salts And Hydrate Nomenclature and then click Get Form to begin.
- Utilize the tools available to fill out your form.
- Emphasize important sections of your documents or redact sensitive information using tools that airSlate SignNow has specifically designed for this purpose.
- Create your electronic signature with the Sign tool, which only takes seconds and holds the same legal validity as a conventional handwritten signature.
- Review all the details and then click the Done button to save your modifications.
- Select how you wish to send your form, whether by email, SMS, or an invitation link, or download it to your computer.
Eliminate concerns about lost or misplaced documents, tedious form searching, or mistakes that necessitate printing new copies. airSlate SignNow addresses all your document management needs in a few clicks from any device you prefer. Modify and eSign Common Cations, Anions, Acids, Salts And Hydrate Nomenclature and guarantee excellent communication at every stage of your form development process with airSlate SignNow.
Create this form in 5 minutes or less
Create this form in 5 minutes!
How to create an eSignature for the common cations anions acids salts and hydrate nomenclature
How to create an electronic signature for a PDF online
How to create an electronic signature for a PDF in Google Chrome
How to create an e-signature for signing PDFs in Gmail
How to create an e-signature right from your smartphone
How to create an e-signature for a PDF on iOS
How to create an e-signature for a PDF on Android
People also ask
-
What are cations and anions in the context of airSlate SignNow's document management?
Cations and anions are types of ions that play a vital role in various chemical reactions, but in the context of airSlate SignNow, they symbolize the interaction between different elements of your document workflow. Understanding how these ions function can help in creating more effective and efficient electronic documents. Our platform streamlines document management, allowing for seamless eSigning and collaboration.
-
How does airSlate SignNow handle the security of documents related to cations and anions?
At airSlate SignNow, we prioritize the security of all documents, including those dealing with cations and anions. Our platform uses advanced encryption protocols and secure data storage to protect sensitive information. This ensures that your documents are not only easily accessible but also safeguarded against unauthorized access.
-
What features does airSlate SignNow offer for managing documents with cations and anions?
airSlate SignNow provides an array of features tailored for effective document management, including templates, custom branding, and real-time collaboration tools. These features are designed to enhance the handling of documents that may involve technical terms like cations and anions, making it easier for your team to work together efficiently.
-
Is airSlate SignNow affordable for small businesses dealing with scientific documents involving cations and anions?
Yes, airSlate SignNow offers competitive pricing plans that are designed to cater to small businesses, especially those dealing with scientific documents, including those that mention cations and anions. Our cost-effective solutions ensure that even small teams can access powerful document management tools without breaking the bank.
-
Can airSlate SignNow integrate with other tools used for managing data on cations and anions?
Absolutely! airSlate SignNow supports integrations with various tools and applications that can enhance your workflow related to cations and anions. This includes CRM software, project management tools, and more, allowing you to create a seamless ecosystem for document handling.
-
What benefits does airSlate SignNow provide when working with documents containing cations and anions?
The benefits of using airSlate SignNow for documents that mention cations and anions include improved efficiency, enhanced collaboration, and secure broadcasting of information. By utilizing our eSigning features, you can quickly execute important documents, reducing turnaround time and streamlining your processes.
-
How easy is it to use airSlate SignNow for cations and anions documentation?
Using airSlate SignNow for managing any documentation, including those that involve cations and anions, is incredibly user-friendly. Our intuitive interface simplifies the process, allowing users to create, send, and sign documents without extensive training, making it accessible for everyone on your team.
Get more for Common Cations, Anions, Acids, Salts And Hydrate Nomenclature
Find out other Common Cations, Anions, Acids, Salts And Hydrate Nomenclature
- How Can I Electronic signature Maine Lawers PPT
- How To Electronic signature Maine Lawers PPT
- Help Me With Electronic signature Minnesota Lawers PDF
- How To Electronic signature Ohio High Tech Presentation
- How Can I Electronic signature Alabama Legal PDF
- How To Electronic signature Alaska Legal Document
- Help Me With Electronic signature Arkansas Legal PDF
- How Can I Electronic signature Arkansas Legal Document
- How Can I Electronic signature California Legal PDF
- Can I Electronic signature Utah High Tech PDF
- How Do I Electronic signature Connecticut Legal Document
- How To Electronic signature Delaware Legal Document
- How Can I Electronic signature Georgia Legal Word
- How Do I Electronic signature Alaska Life Sciences Word
- How Can I Electronic signature Alabama Life Sciences Document
- How Do I Electronic signature Idaho Legal Form
- Help Me With Electronic signature Arizona Life Sciences PDF
- Can I Electronic signature Colorado Non-Profit Form
- How To Electronic signature Indiana Legal Form
- How To Electronic signature Illinois Non-Profit Document