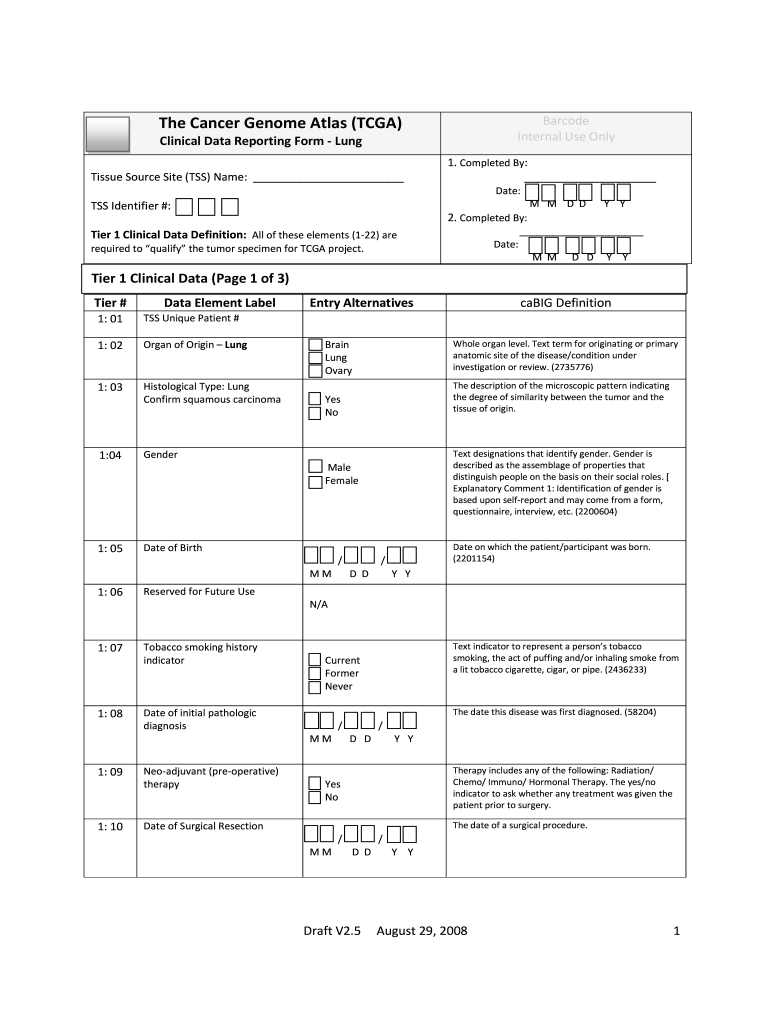
Clinical Data Reporting Form Lung


What is the Clinical Data Reporting Form Lung
The Clinical Data Reporting Form Lung is a specialized document used primarily in the healthcare sector to collect and report data related to lung conditions and treatments. This form is essential for healthcare providers, researchers, and regulatory bodies to ensure accurate tracking of patient outcomes, treatment efficacy, and compliance with clinical guidelines. It serves as a standardized method for gathering critical information that can influence clinical practices and improve patient care.
How to use the Clinical Data Reporting Form Lung
Using the Clinical Data Reporting Form Lung involves several steps to ensure accurate data collection. First, healthcare professionals must familiarize themselves with the form's structure and the specific data points required. This includes patient demographics, clinical history, diagnostic results, and treatment details. Once the relevant information is gathered, it should be entered into the form clearly and accurately. After completion, the form may need to be submitted to a designated authority or database, depending on the regulations governing clinical data reporting in the specific state or institution.
Steps to complete the Clinical Data Reporting Form Lung
Completing the Clinical Data Reporting Form Lung requires careful attention to detail. The following steps can guide users through the process:
- Gather patient information, including name, age, and medical history.
- Document specific lung-related data, such as diagnosis, treatment plans, and outcomes.
- Ensure all sections of the form are filled out completely, avoiding any omissions.
- Review the completed form for accuracy and completeness.
- Submit the form according to the specified guidelines, either electronically or via mail.
Key elements of the Clinical Data Reporting Form Lung
The Clinical Data Reporting Form Lung includes several key elements that are crucial for effective data collection. These elements typically consist of:
- Patient Information: Basic details about the patient, such as name, age, and contact information.
- Clinical History: A summary of the patient's medical background, including previous lung conditions and treatments.
- Diagnostic Information: Results from tests and examinations that confirm the lung condition.
- Treatment Data: Information about the treatments administered, including medications and therapies.
- Outcome Measures: Data reflecting the patient's response to treatment and overall health status.
Legal use of the Clinical Data Reporting Form Lung
The legal use of the Clinical Data Reporting Form Lung is governed by various regulations that ensure patient confidentiality and data integrity. Healthcare providers must comply with the Health Insurance Portability and Accountability Act (HIPAA) when handling patient information. This includes obtaining necessary consent from patients before submitting their data. Additionally, the form must be used in accordance with guidelines set forth by relevant health authorities and organizations to maintain compliance and ensure the validity of the reported data.
Form Submission Methods
The Clinical Data Reporting Form Lung can be submitted through various methods, depending on the requirements of the overseeing body. Common submission methods include:
- Online Submission: Many organizations provide a secure online portal for electronic submission, which streamlines the process and allows for immediate data entry.
- Mail Submission: Users may also opt to print the completed form and send it via postal service to the designated office.
- In-Person Submission: In some cases, submitting the form in person may be required, allowing for direct interaction with the receiving authority.
Quick guide on how to complete clinical data reporting form lung
Easily Prepare Clinical Data Reporting Form Lung on Any Device
Digital document management has gained traction among organizations and individuals alike. It offers an ideal environmentally friendly substitute for traditional printed and signed materials, allowing you to access the correct form and securely store it online. airSlate SignNow provides all the tools necessary to create, edit, and electronically sign your documents quickly and efficiently. Manage Clinical Data Reporting Form Lung on any device using airSlate SignNow's Android or iOS applications and enhance any document-related process today.
How to Edit and Electronically Sign Clinical Data Reporting Form Lung Effortlessly
- Locate Clinical Data Reporting Form Lung and click Get Form to begin.
- Utilize the tools we offer to complete your document.
- Emphasize key sections of the documents or redact sensitive information with the tools that airSlate SignNow provides specifically for that purpose.
- Generate your electronic signature using the Sign tool, which takes mere moments and holds the same legal validity as a conventional handwritten signature.
- Review all the details and then click the Done button to save your changes.
- Choose how you would like to send your form—via email, SMS, or invite link, or download it to your computer.
Say goodbye to lost or misplaced documents, tedious form searching, or errors that necessitate printing new document copies. airSlate SignNow meets your document management needs with just a few clicks from any device you prefer. Edit and electronically sign Clinical Data Reporting Form Lung and ensure effective communication at every stage of the form completion process with airSlate SignNow.
Create this form in 5 minutes or less
Create this form in 5 minutes!
How to create an eSignature for the clinical data reporting form lung
How to create an electronic signature for a PDF online
How to create an electronic signature for a PDF in Google Chrome
How to create an e-signature for signing PDFs in Gmail
How to create an e-signature right from your smartphone
How to create an e-signature for a PDF on iOS
How to create an e-signature for a PDF on Android
People also ask
-
What is a Clinical Data Reporting Form Lung?
A Clinical Data Reporting Form Lung is a specialized document used to collect and report essential clinical information related to lung health and treatments. This form ensures accurate data capture, which is critical for research and patient care. By utilizing the Clinical Data Reporting Form Lung, you can streamline data collection and improve reporting efficiency.
-
How can airSlate SignNow enhance my Clinical Data Reporting Form Lung process?
airSlate SignNow offers a user-friendly platform that allows you to create, send, and eSign your Clinical Data Reporting Form Lung efficiently. The software's features, such as customizable templates and real-time tracking, ensure that your data collection processes are smooth and organized. This leads to better data accuracy and faster turnaround times.
-
What are the pricing options for airSlate SignNow's services?
airSlate SignNow provides flexible pricing plans to meet the needs of different organizations using the Clinical Data Reporting Form Lung. You can choose from subscription plans based on features, storage, and the number of users. Additionally, we offer a free trial, allowing you to explore our services risk-free before making a commitment.
-
Does airSlate SignNow integrate with other software tools?
Yes, airSlate SignNow offers seamless integrations with a variety of applications to enhance your experience with the Clinical Data Reporting Form Lung. You can connect with CRM systems, project management tools, and cloud storage platforms. This allows you to streamline workflows and ensure that all your data is interconnected.
-
What are the benefits of using airSlate SignNow for Clinical Data Reporting Form Lung?
Using airSlate SignNow for your Clinical Data Reporting Form Lung simplifies the document signing process, saves time, and reduces printing costs. The platform ensures compliance with legal requirements through its secure eSignature features. Additionally, you gain access to tracking and analytics, improving oversight of your data collection process.
-
Is airSlate SignNow compliant with healthcare regulations?
Absolutely, airSlate SignNow is designed to meet healthcare compliance standards, ensuring that your Clinical Data Reporting Form Lung adheres to regulations such as HIPAA. Our security measures include data encryption and secure access controls. This gives you peace of mind while managing sensitive clinical data.
-
Can I customize my Clinical Data Reporting Form Lung using airSlate SignNow?
Yes, airSlate SignNow allows you to easily customize your Clinical Data Reporting Form Lung to fit your specific requirements. You can add or remove fields, incorporate branding elements, and adjust the layout to ensure that it meets your organization’s needs. This level of customization streamlines the data collection process.
Get more for Clinical Data Reporting Form Lung
- Images for is it realhttpswwwprudentialcommediamanageddocumentspruannuitiesinvestorord310015pdf form
- How to complete the medicare cms 855i enrollment application form
- What is work related injury and occupational diseaseguide occupational disease and injury reportingwhat is work related injury form
- Fhlbasap federal home loan bank of atlanta form
- We are required by law to obtain information regarding the source of funds and wealth of each planholder and may require this
- Nd8006 source url form
- 2020 form 5498 esa coverdell esa contribution information
- In connection with a business account application form
Find out other Clinical Data Reporting Form Lung
- Sign Rhode Island Tenant lease agreement Myself
- Sign Wyoming Tenant lease agreement Now
- Sign Florida Contract Safe
- Sign Nebraska Contract Safe
- How To Sign North Carolina Contract
- How Can I Sign Alabama Personal loan contract template
- Can I Sign Arizona Personal loan contract template
- How To Sign Arkansas Personal loan contract template
- Sign Colorado Personal loan contract template Mobile
- How Do I Sign Florida Personal loan contract template
- Sign Hawaii Personal loan contract template Safe
- Sign Montana Personal loan contract template Free
- Sign New Mexico Personal loan contract template Myself
- Sign Vermont Real estate contracts Safe
- Can I Sign West Virginia Personal loan contract template
- How Do I Sign Hawaii Real estate sales contract template
- Sign Kentucky New hire forms Myself
- Sign Alabama New hire packet Online
- How Can I Sign California Verification of employment form
- Sign Indiana Home rental application Online