Form CMS 116, CLIA APPLICATION for CERTIFICATION Form CMS 116, CLIA APPLICATION for CERTIFICATION 2021


What is the Form CMS 116?
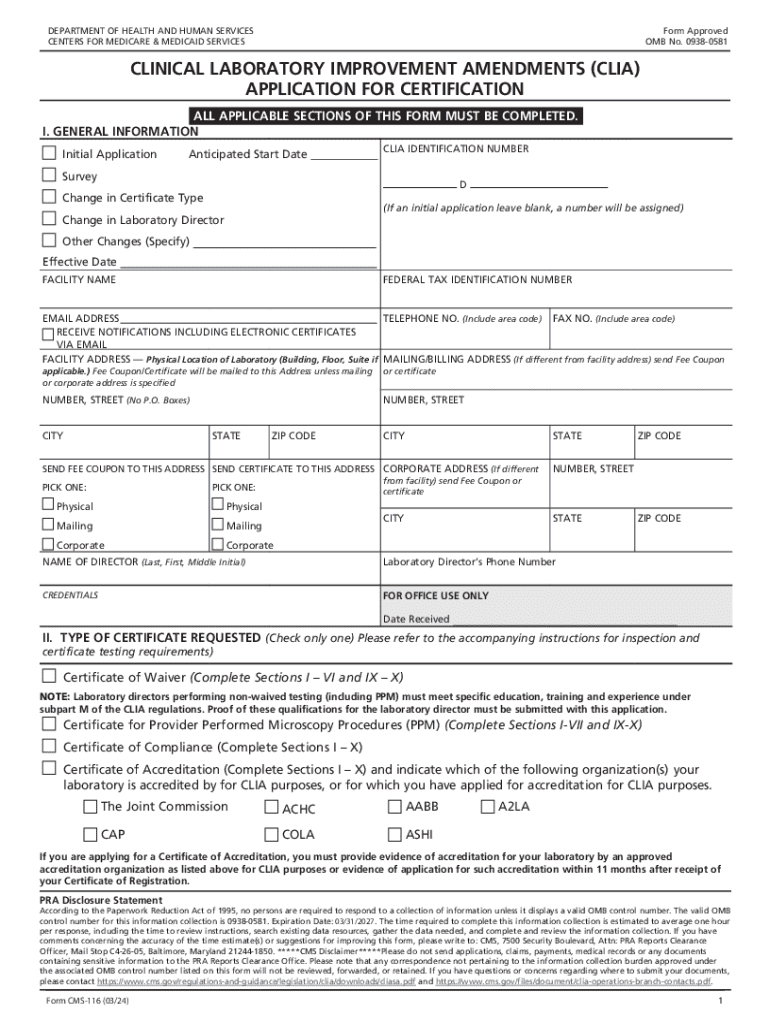
The Form CMS 116, also known as the CLIA Application for Certification, is a crucial document used by laboratories seeking to obtain or renew their Clinical Laboratory Improvement Amendments (CLIA) certification. This form is essential for laboratories that perform tests on human specimens and must comply with federal regulations to ensure the accuracy and reliability of laboratory testing.
This form collects vital information about the laboratory's operations, including its ownership, location, and the types of tests it intends to perform. Completing the CMS 116 is necessary for laboratories to receive a CLIA certificate, which is required to legally operate and bill for laboratory services in the United States.
Steps to Complete the Form CMS 116
Completing the Form CMS 116 involves several key steps to ensure accuracy and compliance with regulatory requirements. Here are the steps to follow:
- Gather Required Information: Collect all necessary data, including the laboratory's name, address, and ownership details.
- Identify Test Categories: Determine the specific categories of tests the laboratory will perform, as this affects the type of CLIA certificate needed.
- Complete the Form: Fill out the form carefully, ensuring all sections are completed accurately. Pay attention to any specific instructions provided on the form.
- Review for Accuracy: Double-check all entries for completeness and correctness to avoid delays in processing.
- Submit the Form: Follow the submission guidelines for the CMS 116, which may include online submission or mailing the completed form to the appropriate state agency.
How to Obtain the Form CMS 116
The Form CMS 116 can be obtained through various channels to ensure accessibility for all laboratories. Here are the primary ways to acquire this form:
- Online Download: Visit the official Centers for Medicare & Medicaid Services (CMS) website to download the form directly in PDF format.
- Request by Mail: Laboratories can request a physical copy of the form by contacting their state health department or the CMS regional office.
- Local Health Departments: Many local health departments may have copies of the form available for distribution.
Required Documents for Form CMS 116 Submission
When submitting the Form CMS 116, laboratories must include several documents to support their application. These documents may include:
- Proof of Ownership: Documentation that verifies the ownership structure of the laboratory.
- Facility License: A copy of any applicable state licenses or permits required for laboratory operation.
- Test Menu: A detailed list of all tests the laboratory intends to perform, categorized by complexity.
Eligibility Criteria for CLIA Certification
To be eligible for CLIA certification, laboratories must meet specific criteria outlined by the CMS. These criteria include:
- Type of Laboratory: The laboratory must be a facility that performs testing on human specimens.
- Compliance with Regulations: The laboratory must agree to comply with all applicable federal and state regulations governing laboratory testing.
- Quality Assurance Programs: Laboratories must implement quality assurance and quality control programs to maintain testing accuracy.
Form Submission Methods
Laboratories can submit the Form CMS 116 through various methods, ensuring flexibility and convenience. The submission options include:
- Online Submission: Many states allow for the electronic submission of the form through their health department websites.
- Mail Submission: Laboratories can print the completed form and mail it to the designated state agency or CMS regional office.
- In-Person Submission: Some laboratories may choose to submit the form in person at their local health department or CMS office.
Quick guide on how to complete form cms 116 clia application for certification form cms 116 clia application for certification
Create Form CMS 116, CLIA APPLICATION FOR CERTIFICATION Form CMS 116, CLIA APPLICATION FOR CERTIFICATION effortlessly on any device
Digital document management has become increasingly favored by businesses and individuals alike. It serves as an ideal environmentally friendly alternative to traditional printed and signed papers, allowing you to locate the correct form and securely keep it online. airSlate SignNow equips you with all the necessary tools to create, modify, and eSign your documents quickly without delays. Handle Form CMS 116, CLIA APPLICATION FOR CERTIFICATION Form CMS 116, CLIA APPLICATION FOR CERTIFICATION on any device with airSlate SignNow Android or iOS applications and enhance any document-focused procedure today.
Steps to modify and eSign Form CMS 116, CLIA APPLICATION FOR CERTIFICATION Form CMS 116, CLIA APPLICATION FOR CERTIFICATION seamlessly
- Locate Form CMS 116, CLIA APPLICATION FOR CERTIFICATION Form CMS 116, CLIA APPLICATION FOR CERTIFICATION and click Get Form to begin.
- Utilize the features we offer to complete your form.
- Emphasize pertinent sections of your documents or obscure sensitive information with tools that airSlate SignNow specifically provides for this purpose.
- Generate your eSignature using the Sign tool, which takes mere seconds and carries the same legal validity as a conventional wet ink signature.
- Review the details and click on the Done button to save your changes.
- Choose how you wish to share your form, via email, text message (SMS), or invitation link, or download it to your computer.
Eliminate worries about lost or misplaced files, tedious form searches, or errors that necessitate printing new document versions. airSlate SignNow addresses all your document management needs in just a few clicks from any device you prefer. Modify and eSign Form CMS 116, CLIA APPLICATION FOR CERTIFICATION Form CMS 116, CLIA APPLICATION FOR CERTIFICATION to ensure excellent communication at every stage of the document preparation process with airSlate SignNow.
Create this form in 5 minutes or less
Find and fill out the correct form cms 116 clia application for certification form cms 116 clia application for certification
Create this form in 5 minutes!
How to create an eSignature for the form cms 116 clia application for certification form cms 116 clia application for certification
How to create an electronic signature for a PDF online
How to create an electronic signature for a PDF in Google Chrome
How to create an e-signature for signing PDFs in Gmail
How to create an e-signature right from your smartphone
How to create an e-signature for a PDF on iOS
How to create an e-signature for a PDF on Android
People also ask
-
What is health human services CLIA and how does it relate to airSlate SignNow?
Health human services CLIA refers to the Clinical Laboratory Improvement Amendments, which set standards for laboratory testing. airSlate SignNow provides a secure platform for eSigning documents related to health human services CLIA compliance, ensuring that your documents are legally binding and easily accessible.
-
How can airSlate SignNow help with CLIA compliance in health human services?
airSlate SignNow streamlines the document signing process, making it easier to manage compliance with health human services CLIA regulations. By using our platform, you can ensure that all necessary documents are signed and stored securely, reducing the risk of non-compliance.
-
What features does airSlate SignNow offer for health human services CLIA documentation?
Our platform offers features such as customizable templates, automated workflows, and secure storage, all tailored for health human services CLIA documentation. These features help you efficiently manage your documents while ensuring compliance with regulatory standards.
-
Is airSlate SignNow cost-effective for businesses in the health human services sector?
Yes, airSlate SignNow is designed to be a cost-effective solution for businesses in the health human services sector. With flexible pricing plans, you can choose the option that best fits your needs while ensuring compliance with health human services CLIA requirements.
-
Can airSlate SignNow integrate with other tools used in health human services?
Absolutely! airSlate SignNow offers integrations with various tools commonly used in health human services, enhancing your workflow efficiency. This allows you to seamlessly manage your documents while adhering to health human services CLIA standards.
-
What are the benefits of using airSlate SignNow for health human services CLIA documentation?
Using airSlate SignNow for health human services CLIA documentation provides numerous benefits, including improved efficiency, enhanced security, and better compliance management. Our platform simplifies the signing process, allowing you to focus on delivering quality services.
-
How does airSlate SignNow ensure the security of health human services CLIA documents?
airSlate SignNow employs advanced security measures, including encryption and secure access controls, to protect your health human services CLIA documents. This ensures that sensitive information remains confidential and compliant with industry regulations.
Get more for Form CMS 116, CLIA APPLICATION FOR CERTIFICATION Form CMS 116, CLIA APPLICATION FOR CERTIFICATION
Find out other Form CMS 116, CLIA APPLICATION FOR CERTIFICATION Form CMS 116, CLIA APPLICATION FOR CERTIFICATION
- Can I eSignature Massachusetts Real Estate PDF
- How Can I eSignature New Jersey Police Document
- How Can I eSignature New Jersey Real Estate Word
- Can I eSignature Tennessee Police Form
- How Can I eSignature Vermont Police Presentation
- How Do I eSignature Pennsylvania Real Estate Document
- How Do I eSignature Texas Real Estate Document
- How Can I eSignature Colorado Courts PDF
- Can I eSignature Louisiana Courts Document
- How To Electronic signature Arkansas Banking Document
- How Do I Electronic signature California Banking Form
- How Do I eSignature Michigan Courts Document
- Can I eSignature Missouri Courts Document
- How Can I Electronic signature Delaware Banking PDF
- Can I Electronic signature Hawaii Banking Document
- Can I eSignature North Carolina Courts Presentation
- Can I eSignature Oklahoma Courts Word
- How To Electronic signature Alabama Business Operations Form
- Help Me With Electronic signature Alabama Car Dealer Presentation
- How Can I Electronic signature California Car Dealer PDF