Fda 1572 2019-2026


What is the FDA 1572?
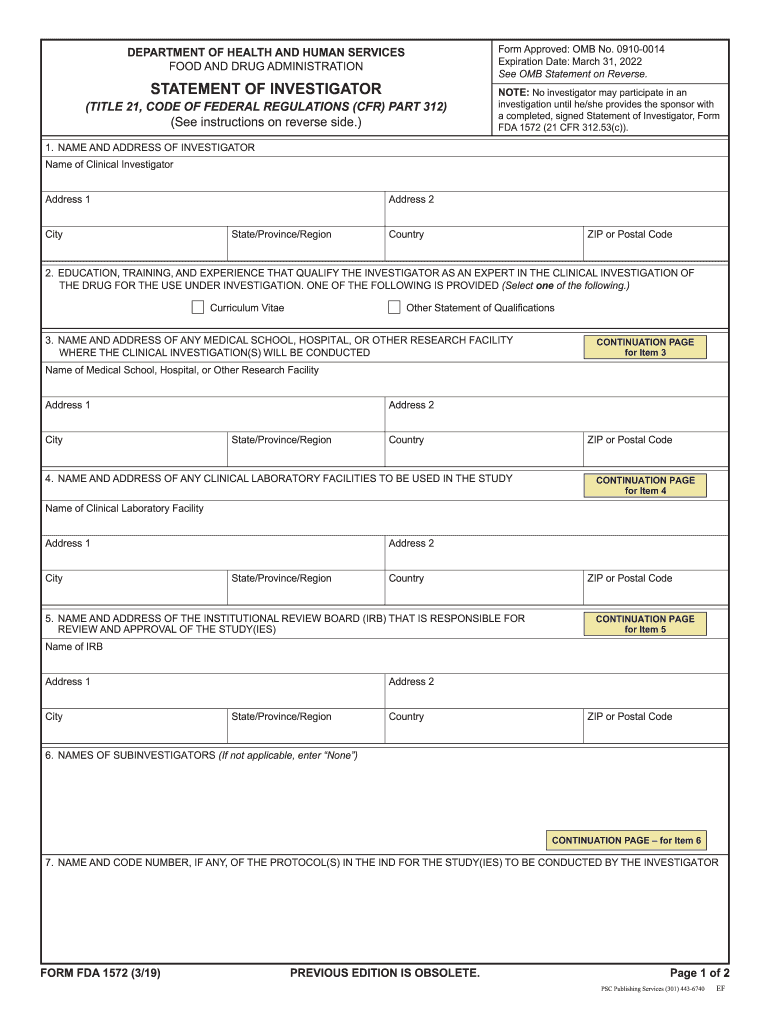
The FDA 1572 form, also known as the Investigator's Statement, is a critical document required by the Food and Drug Administration (FDA) for clinical trials involving investigational drugs. This form serves as a declaration by the investigator, affirming their qualifications and commitment to conducting the trial in compliance with regulatory requirements. It includes essential information such as the investigator's name, address, and qualifications, as well as details about the study site and the study protocol. The FDA 1572 is vital for ensuring that trials adhere to ethical and scientific standards.
Steps to Complete the FDA 1572
Completing the FDA 1572 form involves several key steps to ensure accuracy and compliance. First, gather all necessary information, including your credentials and details about the clinical study. Next, fill out the form by providing your name, contact information, and the names of any sub-investigators. It is also important to include the study title and protocol number. After completing the form, review it carefully for any errors or omissions. Finally, sign and date the form to validate your commitment to the study's compliance with FDA regulations.
Legal Use of the FDA 1572
The legal use of the FDA 1572 form is governed by various regulations that ensure the integrity of clinical trials. This form must be completed accurately and submitted to the FDA as part of the Investigational New Drug (IND) application process. Compliance with the regulations outlined in the form is essential for the legal execution of clinical studies. Failure to adhere to these requirements can result in penalties, including the rejection of the IND application or other legal repercussions. Therefore, understanding the legal implications of the FDA 1572 is crucial for investigators.
Key Elements of the FDA 1572
The FDA 1572 form includes several key elements that are essential for its validity. These elements include the investigator's professional qualifications, the study's title and protocol number, and the location where the study will be conducted. Additionally, the form requires information about any sub-investigators and their roles in the study. The acknowledgment of the investigator's responsibilities, including adherence to Good Clinical Practice (GCP), is also a critical component. Each of these elements plays a vital role in ensuring that the clinical trial is conducted ethically and responsibly.
How to Use the FDA 1572
Using the FDA 1572 form effectively requires understanding its purpose and the context in which it is utilized. The form is primarily used during the initiation of clinical trials to inform the FDA about the investigator's qualifications and the study's details. Once completed, it should be submitted as part of the IND application, along with other required documents. Throughout the study, the investigator must ensure that any changes to the study or personnel are documented and communicated to the FDA, maintaining compliance with regulatory standards.
Form Submission Methods
The FDA 1572 form can be submitted through various methods, depending on the requirements of the specific clinical trial. Typically, the form is submitted electronically as part of the Investigational New Drug (IND) application, which allows for faster processing and easier tracking. In some cases, the form may also be submitted via mail or in person, particularly if there are specific instructions from the FDA or the sponsoring organization. It is important to follow the submission guidelines provided by the FDA to ensure that the form is processed without delays.
Quick guide on how to complete expiration date march 31 2022
Effortlessly Prepare Fda 1572 on Any Device
Digital document management has gained traction among businesses and individuals alike. It serves as an excellent eco-friendly substitute for conventional printed and signed papers, allowing you to obtain the appropriate form and securely save it online. airSlate SignNow provides you with all the tools necessary to create, modify, and electronically sign your documents promptly without delays. Manage Fda 1572 on any platform using airSlate SignNow Android or iOS applications and streamline any document-based task today.
How to Modify and Electronically Sign Fda 1572 with Ease
- Find Fda 1572 and click Get Form to begin.
- Utilize the tools we provide to complete your form.
- Highlight pertinent sections of your documents or obscure sensitive data with tools specifically offered by airSlate SignNow for this purpose.
- Create your signature using the Sign tool, which takes mere seconds and holds the same legal validity as a traditional wet ink signature.
- Review all the information and click on the Done button to save your changes.
- Choose your preferred method to send your form—by email, SMS, invite link, or download it to your computer.
Eliminate the hassle of lost or misplaced files, tedious form navigation, or mistakes that necessitate printing new document copies. airSlate SignNow addresses all your document management needs in just a few clicks from your chosen device. Edit and electronically sign Fda 1572 to ensure seamless communication at any stage of the form preparation process with airSlate SignNow.
Create this form in 5 minutes or less
Find and fill out the correct expiration date march 31 2022
Create this form in 5 minutes!
How to create an eSignature for the expiration date march 31 2022
How to generate an electronic signature for the Expiration Date March 31 2022 online
How to create an electronic signature for the Expiration Date March 31 2022 in Google Chrome
How to generate an eSignature for putting it on the Expiration Date March 31 2022 in Gmail
How to create an eSignature for the Expiration Date March 31 2022 straight from your mobile device
How to create an electronic signature for the Expiration Date March 31 2022 on iOS
How to create an electronic signature for the Expiration Date March 31 2022 on Android
People also ask
-
What is the Fda 1572 form and why is it important?
The Fda 1572 form, also known as the Statement of Investigator, is a critical document used in clinical trials to ensure compliance with FDA regulations. It outlines the responsibilities of investigators and provides essential information about the study. Understanding and properly completing the Fda 1572 form is crucial for successful trial management.
-
How can airSlate SignNow help with the Fda 1572 form?
airSlate SignNow simplifies the process of completing and signing the Fda 1572 form by providing a secure and user-friendly platform. With features like eSignature and document templates, you can easily manage and track your submissions. This streamlines compliance and ensures that your documentation meets all regulatory requirements.
-
Is airSlate SignNow cost-effective for handling the Fda 1572 form?
Yes, airSlate SignNow offers a cost-effective solution for managing the Fda 1572 form and other essential documents. Our pricing plans are designed to fit businesses of all sizes, providing value without compromising on features. This allows you to focus more on your clinical trials while keeping costs manageable.
-
What features does airSlate SignNow offer for the Fda 1572 form?
airSlate SignNow provides a variety of features specifically designed to assist with the Fda 1572 form, including customizable templates, eSigning capabilities, and secure cloud storage. Additionally, our platform ensures that all documents are legally binding and compliant with FDA regulations, making it easier to manage your trial documentation.
-
Can I integrate airSlate SignNow with other software for the Fda 1572 process?
Absolutely! airSlate SignNow offers seamless integrations with various software solutions that can enhance your workflow for the Fda 1572 form. Whether you’re using project management tools or document management systems, our integrations help streamline your processes and improve efficiency.
-
What are the benefits of using airSlate SignNow for clinical trial documentation like the Fda 1572?
Using airSlate SignNow for clinical trial documentation, including the Fda 1572 form, offers numerous benefits. You gain access to a secure, efficient, and compliant platform that simplifies the signing process. This not only speeds up document handling but also enhances collaboration among team members and investigators.
-
How does airSlate SignNow ensure the security of the Fda 1572 form?
airSlate SignNow prioritizes security, especially when handling sensitive documents like the Fda 1572 form. Our platform utilizes advanced encryption methods, secure cloud storage, and strict access controls to protect your data. This ensures that your clinical trial documentation remains confidential and secure throughout the process.
Get more for Fda 1572
- Printresetpurchasing agent appointmentform17and de
- 11288b request under section 88b of the inland revenue ordinance cap 112 for a notice of no objection to a company being form
- Personal injury application connecticut judicial branch form
- Initial completion date form
- Ml application form doc
- Adult family home initial licensure checklist f 62671 1100 form
- Fillable online toastmasters form 3 charter membership
- Indiabulls account closure form fill out ampamp sign online
Find out other Fda 1572
- Sign Montana Lawers LLC Operating Agreement Free
- Sign Montana Lawers LLC Operating Agreement Fast
- Can I Sign Nevada Lawers Letter Of Intent
- Sign Minnesota Insurance Residential Lease Agreement Fast
- How Do I Sign Ohio Lawers LLC Operating Agreement
- Sign Oregon Lawers Limited Power Of Attorney Simple
- Sign Oregon Lawers POA Online
- Sign Mississippi Insurance POA Fast
- How Do I Sign South Carolina Lawers Limited Power Of Attorney
- Sign South Dakota Lawers Quitclaim Deed Fast
- Sign South Dakota Lawers Memorandum Of Understanding Free
- Sign South Dakota Lawers Limited Power Of Attorney Now
- Sign Texas Lawers Limited Power Of Attorney Safe
- Sign Tennessee Lawers Affidavit Of Heirship Free
- Sign Vermont Lawers Quitclaim Deed Simple
- Sign Vermont Lawers Cease And Desist Letter Free
- Sign Nevada Insurance Lease Agreement Mobile
- Can I Sign Washington Lawers Quitclaim Deed
- Sign West Virginia Lawers Arbitration Agreement Secure
- Sign Wyoming Lawers Lease Agreement Now