Routine Vaccine Storage and Handling Plan Worksheet Form


What is the Routine Vaccine Storage And Handling Plan Worksheet
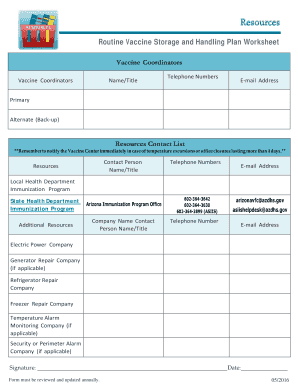
The Routine Vaccine Storage and Handling Plan Worksheet is a crucial document designed to assist healthcare providers in maintaining the integrity of vaccines. This worksheet outlines best practices for storing and handling vaccines, ensuring they remain effective and safe for patient use. It serves as a guideline for proper temperature monitoring, storage conditions, and inventory management, which are essential in preventing vaccine spoilage and ensuring compliance with health regulations.
How to use the Routine Vaccine Storage And Handling Plan Worksheet
Using the Routine Vaccine Storage and Handling Plan Worksheet involves several steps. First, healthcare providers should familiarize themselves with the worksheet's sections, which cover various aspects of vaccine management. Next, they should regularly fill out the worksheet to document storage conditions, including temperature logs and inventory counts. This ongoing documentation helps identify any issues promptly and ensures adherence to recommended practices. Regular reviews of the completed worksheet can also aid in staff training and compliance audits.
Key elements of the Routine Vaccine Storage And Handling Plan Worksheet
The key elements of the Routine Vaccine Storage and Handling Plan Worksheet include:
- Temperature Monitoring: Regularly recording the temperatures of storage units to ensure they remain within the recommended range.
- Inventory Management: Keeping an accurate count of vaccine stock, including expiration dates and lot numbers.
- Staff Training: Documenting training sessions for staff on proper vaccine handling and storage protocols.
- Emergency Procedures: Outlining steps to take in case of temperature excursions or equipment failures.
Steps to complete the Routine Vaccine Storage And Handling Plan Worksheet
Completing the Routine Vaccine Storage and Handling Plan Worksheet involves a systematic approach:
- Gather necessary materials, including temperature logs and inventory lists.
- Record the current temperature of the vaccine storage unit.
- Update the inventory section with the current stock and expiration dates.
- Review and document staff training sessions related to vaccine storage.
- Outline any incidents or deviations from standard procedures and the corrective actions taken.
Legal use of the Routine Vaccine Storage And Handling Plan Worksheet
The Routine Vaccine Storage and Handling Plan Worksheet is legally significant as it supports compliance with federal and state regulations regarding vaccine storage. Proper documentation can serve as evidence of adherence to guidelines set forth by health authorities, such as the Centers for Disease Control and Prevention (CDC). In case of audits or investigations, having a well-maintained worksheet can demonstrate a commitment to patient safety and regulatory compliance.
State-specific rules for the Routine Vaccine Storage And Handling Plan Worksheet
State-specific regulations may dictate additional requirements for the Routine Vaccine Storage and Handling Plan Worksheet. Healthcare providers should be aware of their state's guidelines regarding vaccine storage, reporting requirements, and any specific documentation needed. This ensures that they not only comply with federal standards but also adhere to local laws that may impose stricter regulations on vaccine management.
Quick guide on how to complete routine vaccine storage and handling plan worksheet
Prepare [SKS] effortlessly on any device
Digital document management has gained signNow traction among organizations and individuals. It serves as an ideal eco-friendly alternative to conventional printed and signed papers, enabling you to locate the correct form and securely store it online. airSlate SignNow provides all the tools necessary to create, modify, and eSign your documents quickly without delays. Manage [SKS] on any platform with the airSlate SignNow Android or iOS applications and enhance any document-oriented process today.
The easiest way to modify and eSign [SKS] with ease
- Locate [SKS] and click Get Form to begin.
- Utilize the tools we provide to fill out your form.
- Highlight pertinent sections of the documents or obscure sensitive details with tools that airSlate SignNow offers specifically for that purpose.
- Have your eSignature created with the Sign feature, which takes mere seconds and holds the same legal validity as a traditional handwritten signature.
- Review the information and click the Done button to save your modifications.
- Select your preferred method of sending your form, whether by email, text message (SMS), invite link, or download it to your computer.
Eliminate concerns about lost or misplaced documents, tedious form searching, or errors that necessitate printing new document copies. airSlate SignNow manages all your document management needs in just a few clicks from any device of your choice. Adjust and eSign [SKS] to ensure outstanding communication at every step of your form preparation process with airSlate SignNow.
Create this form in 5 minutes or less
Related searches to Routine Vaccine Storage And Handling Plan Worksheet
Create this form in 5 minutes!
How to create an eSignature for the routine vaccine storage and handling plan worksheet
How to create an electronic signature for a PDF online
How to create an electronic signature for a PDF in Google Chrome
How to create an e-signature for signing PDFs in Gmail
How to create an e-signature right from your smartphone
How to create an e-signature for a PDF on iOS
How to create an e-signature for a PDF on Android
People also ask
-
What is a Routine Vaccine Storage And Handling Plan Worksheet?
A Routine Vaccine Storage And Handling Plan Worksheet is a vital tool that outlines the proper procedures and protocols for storing and handling vaccines safely. It ensures that vaccines maintain their efficacy and are stored at the correct temperatures. This worksheet is essential for healthcare providers to comply with regulatory guidelines.
-
How can the Routine Vaccine Storage And Handling Plan Worksheet benefit my practice?
Using the Routine Vaccine Storage And Handling Plan Worksheet can signNowly enhance your practice's vaccine management. It helps minimize the risk of improper storage, ensuring vaccines remain viable and effective. Additionally, it can streamline the training of staff on storage protocols, enhancing overall compliance.
-
Is the Routine Vaccine Storage And Handling Plan Worksheet customizable?
Yes, the Routine Vaccine Storage And Handling Plan Worksheet can be tailored to meet the specific needs of your organization. This flexibility allows you to incorporate unique protocols and procedures relevant to your practice or facility. Customization ensures that all team members are aligned on vaccine handling standards.
-
What features does the airSlate SignNow platform offer for managing vaccine-related documents?
The airSlate SignNow platform provides numerous features including e-signature capabilities, document templates, and secure document storage. These tools support the effective management of the Routine Vaccine Storage And Handling Plan Worksheet. Simplifying document workflows enhances efficiency and ensures compliance with vaccination protocols.
-
How does airSlate SignNow integrate with other health record systems for vaccine management?
airSlate SignNow seamlessly integrates with various health record systems, allowing for efficient sharing and management of vaccine-related documents. This compatibility ensures that the Routine Vaccine Storage And Handling Plan Worksheet can easily be incorporated into your existing workflow. Integration simplifies documentation and record-keeping related to vaccinations.
-
What kind of support does airSlate SignNow provide for users of the Routine Vaccine Storage And Handling Plan Worksheet?
Users of the Routine Vaccine Storage And Handling Plan Worksheet through airSlate SignNow benefit from comprehensive customer support. This includes access to tutorials, user guides, and a responsive help center to address any queries. Our support ensures that you can maximize the platform's features and effectively manage your vaccine documentation.
-
Is there a free trial available for airSlate SignNow?
Yes, airSlate SignNow offers a free trial period that allows you to explore the features related to the Routine Vaccine Storage And Handling Plan Worksheet. This trial enables you to assess how well the solution fits your needs without any upfront commitment. Take advantage of this opportunity to enhance your vaccine management processes.
Get more for Routine Vaccine Storage And Handling Plan Worksheet
Find out other Routine Vaccine Storage And Handling Plan Worksheet
- Can I eSign South Carolina Real estate contracts
- eSign Texas Renter's contract Mobile
- How Do I eSign Texas Renter's contract
- eSign Hawaii Sales contract template Myself
- How Can I eSign Washington Real estate sales contract template
- How To eSignature California Stock Certificate
- How Can I eSignature Texas Stock Certificate
- Help Me With eSign Florida New employee checklist
- How To eSign Illinois Rental application
- How To eSignature Maryland Affidavit of Identity
- eSignature New York Affidavit of Service Easy
- How To eSignature Idaho Affidavit of Title
- eSign Wisconsin Real estate forms Secure
- How To eSign California Real estate investment proposal template
- eSignature Oregon Affidavit of Title Free
- eSign Colorado Real estate investment proposal template Simple
- eSign Louisiana Real estate investment proposal template Fast
- eSign Wyoming Real estate investment proposal template Free
- How Can I eSign New York Residential lease
- eSignature Colorado Cease and Desist Letter Later