eSign Protocol Made Easy
Get the robust eSignature features you need from the solution you trust
Choose the pro platform created for professionals
Configure eSignature API with ease
Work better together
Esign protocol, within a few minutes
Cut the closing time
Maintain important data safe
See airSlate SignNow eSignatures in action
airSlate SignNow solutions for better efficiency
Our user reviews speak for themselves






Why choose airSlate SignNow
-
Free 7-day trial. Choose the plan you need and try it risk-free.
-
Honest pricing for full-featured plans. airSlate SignNow offers subscription plans with no overages or hidden fees at renewal.
-
Enterprise-grade security. airSlate SignNow helps you comply with global security standards.

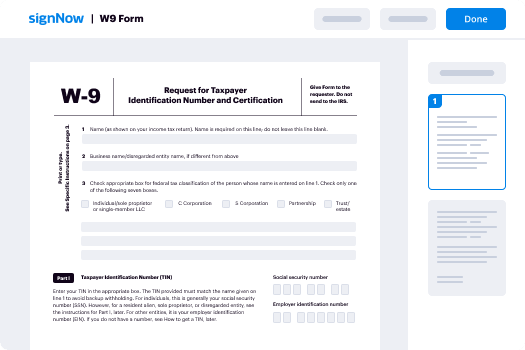
Your step-by-step guide — esign protocol
Using airSlate SignNow’s electronic signature any organization can increase signature workflows and eSign in real-time, delivering an improved experience to clients and staff members. Use esign protocol in a few simple actions. Our handheld mobile apps make operating on the go feasible, even while offline! Sign signNows from any place worldwide and complete deals in less time.
Keep to the walk-through instruction for using esign protocol:
- Sign in to your airSlate SignNow profile.
- Locate your record in your folders or upload a new one.
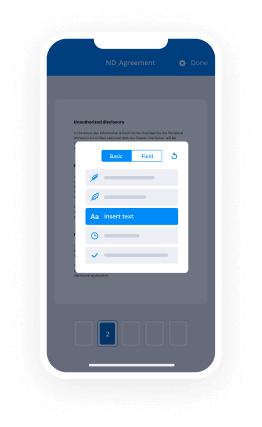
- Open up the record adjust using the Tools list.
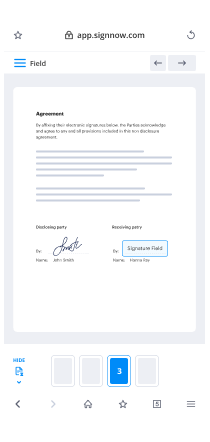
- Drop fillable fields, add textual content and sign it.
- List multiple signers by emails configure the signing sequence.
- Indicate which users can get an signed version.
- Use Advanced Options to limit access to the template and set an expiry date.
- Press Save and Close when done.
Moreover, there are more advanced capabilities accessible for esign protocol. List users to your shared workspace, view teams, and monitor teamwork. Numerous people all over the US and Europe recognize that a solution that brings people together in a single holistic digital location, is the thing that organizations need to keep workflows performing effortlessly. The airSlate SignNow REST API enables you to integrate eSignatures into your app, internet site, CRM or cloud storage. Check out airSlate SignNow and enjoy quicker, smoother and overall more effective eSignature workflows!
How it works
airSlate SignNow features that users love
See exceptional results esign protocol made easy
Get legally-binding signatures now!
FAQs
-
How do I electronically sign a document?
Windows: Use airSlate SignNow Reader To sign a document using airSlate SignNow Reader, first open the PDF document in the airSlate SignNow Reader DC application. Click the \u201cFill & Sign\u201d button in the right pane. Click the \u201cSign\u201d button on the toolbar and select \u201cAdd Signature\u201d to add your signature to airSlate SignNow Reader DC. -
How do I send an electronic signature?
Open a PDF file in Acrobat DC. Click the Fill & Sign tool in the right pane. Add a recipient: Enter an email address and add a custom message if you want. ... Create your form and signature fields: ... Send your form: -
How do I do an electronic signature in Word?
To add a digital signature, open your Microsoft Word document and click where you'd like to add your signature line. From the Word ribbon, select the Insert tab and then click Signature Line in the Text group. A Signature Setup pop-up box appears. Enter your information in the text fields and click OK. -
How can I eSign for free?
Choose a File to Sign. Choose the document you want to have electronically signed online. ... Set Signer Details. Register the signer's name and email address. ... Send for Signature. Your signer will receive an email requesting their signature. ... Sign and Download. -
How do I electronically sign a form?
Windows: Use airSlate SignNow Reader To sign a document using airSlate SignNow Reader, first open the PDF document in the airSlate SignNow Reader DC application. Click the \u201cFill & Sign\u201d button in the right pane. Click the \u201cSign\u201d button on the toolbar and select \u201cAdd Signature\u201d to add your signature to airSlate SignNow Reader DC. -
How do I electronically sign an online form?
Locate the signature section of the online application. For online applications that insert the signature you create, you generally select \u201csign document\u201d or a similar instruction and then click \u201cokay\u201d or \u201capply signature\u201d to complete the process. -
How do you convert a PDF to a fillable form?
Step 1: Load the PDF File. Open your pre-existing PDF form in PDFelement, or you can choose to open an existing form template. ... Step 2: Recognize Form Fields Automatically. ... Step 3: Fill and Save the Form. -
What does it mean to electronically sign a document?
What is an electronic signature? An electronic signature is a symbol or other data in digital format that is attached to an electronic document and applied or adopted by a person with an intent to sign. Most often, eSignatures take the form of an image of a physical signature. -
How can I create a digital signature for free?
Suggested clip Create a Free Digital Signature for Signing PDFs - YouTubeYouTubeStart of suggested clipEnd of suggested clip Create a Free Digital Signature for Signing PDFs - YouTube -
What is E sign consent?
The eSign Act allows the use of electronic records to satisfy any statute, regulation, or rule of law requiring that such information be provided in writing, if the consumer has affirmatively consented to such use and has not withdrawn such consent. -
What is an electronic signature and how does it work?
Electronic signatures\u2014also called eSignatures\u2014are any sound, symbol, or process that shows the intent to sign something. This could be a scan of your hand-written signature, a stamp, or a recorded verbal confirmation. An electronic signature could even be your typed name on the signature line of a document. -
What federal act allowed the legal use of e signatures?
The ESIGN Act is a federal law passed in 2000. It grants legal recognition to electronic signatures and records if all parties to a contract choose to use electronic documents and to sign them electronically. -
How do I get an electronic signature?
In the Tools Center, select Prepare Form. Choose Select a file and browse to open the file that needs signing. ... Form and signature fields are automatically added. ... Click Ready to Send and enter the recipients email addresses. ... Check your email to confirm that the agreement has been sent and to track the progress. -
What is meant by eSign?
eSign is an online electronic signature service that can facilitate an Aadhaar holder to digitally sign a document. An Aadhaar holder can now sign a document after Biometric/One Time Password authentication thus requiring no airSlate SignNow based application form or documents.
What active users are saying — esign protocol
Related searches to esign protocol made easy
Esign protocol
hi this is Rob Packard the medical device Academy this is a brief video explaining what you get when you purchase our design control procedure sys 0:08 this email will come to you automatically after you confirm your desire to subscribe to our automated emails giving you updates to our procedures free the only attachment to this is a design review meeting minutes form there are some the combined file size of the other documents is too large so we are providing hyperlinks to download them from our Dropbox folder so here are the different hyperlinks there's the procedure there's a design transfer checklist a traceability matrix a design and risk management plan that's combined and then there's a bunch of templates for user nior product requirements user needs inputs verification protocol valid verification report and a risk management report template now I'll show you each of those documents so this this procedure has gotten much longer than it was before because we've added quite a bit more detail this is the design control procedure it's now up to 14 pages in this procedure we've listed all the different templates that we are that are included in this Matthew Walker has been very busy adding hyperlinks in color coding he's added in blue color coding for each of the clauses in the standard so it's easy for auditors to find all the regulatory requirements then when you go further down here we explain what things we added throughout the procedure you're gonna see more of these blue references to the different ISO 1345 2016 clauses and we've added a lot more references to the different templates so here's a one of our forums there's a template here's another template and then throughout this we have more blue hyperlinks and more references to templates and then when you go down to the bottom of the procedure and this is all organized by phases so this is stage 4 and then we have stage 5 the final stage for product release and after you get through that it talks about monitoring and measuring what things to monitor in the design process you can modify this to measure the things that you want to measure for your own quality objectives it says who's responsible for the process and what training they need to go through talks about risk management for the process because we use a risk-based approach and all the procedures and it talks about the records and here all the different records that you would put in a design history file organized by phase and this is very much a copy from what's also in the design plan so for each part we've just recently added all the cross references to the different templates to help people keep track of where which documents go where and which sections and then at the end we have our diagram showing the design process this is a combination showing the typical phase Gate approach plus all the risk management activities that would occur and then major milestones in a typical 510 case mission but these could be replaced with European CE Marking requirements as well which would be a little bit shifted out because you can you need to submit clinical data for CE Marking submission is one of the templates so here's a verification report template this is created by Mary Vader she's got in here some software references but this is a very simplified report or template you would add more things in here we have other templates that have more detail if you need them the design review meeting minutes this would be the same review meeting minutes form that you would use for all the design reviews we don't have a separate one for each review we use the same template for each and then you would indicate wouldn't be updated before you go into the next phase this is the design transfer checklist we created this recently to help companies keep track of all the things they need to do during the design transfer process so the person responsible for each of these milestones to make sure they were all completed the target date for completing them in the design transfer and then the data was actually completed so we've got 18 different items here and we have included links to templates as well next item is the design risk management plan template this is a device description that you would put in here that would be the same as what you would use for your designed this device subscription in a 510 K summary just the initial paragraph the general description then we have an overview of the project and then we've broken out each of those boxes as separate Bock tables for each phase and still we have these links for the different documents in here and then we have explanations about what procedures apply and what goes on in between the phases in between each of those tables and see the procedure references we referenced the design procedure here the risk management procedure and the usability procedure and then we say who's responsible for this at the end the next requirement or next template is the risk management report this is in here because we have both the risk management is integrated with the design process we provided this template as well we also provide this as part of our purchase in the risk management procedure and it has the different things that are required at each of the phases and next we have design inputs document I think this is one way to describe it with a lot more detail another way is to use our traceability matrix so we've got a combined design requirements traceability matrix and you could put design inputs here you could also put hazards or if you need more room you could just reference the document where it can be found and what line item it is and then you'd put it in that other document here and indicate what ID it is so design input number one two three four so you'd indicate which one it is and would say it's in template 24 or whatever document number you give it this here is sort of a combination of your input output verification validation io v V as well as the hazard risk management documents that you would need so user needs hazards inputs risk controls severity of harm probability of occurrence of harm or probability of occurrence that's P 1 it's a P 2 over here that you would determine after you gather some clinical data and residual risk that there's a place to put residual risk that you would link in your warnings of precautions in your ifu and then anything that you link for post marco clinical follow-up studies and reports a user needs document here this is a template and once again we have traceability with numbers and you ideally want to have these numbers linked with the inputs and outputs hazards we also have a product requirements document this would also be another user needs type document but it he added even higher level this is more from a business standpoint and last but not least we have the verification protocol template so this says these are the protocol leading into the report that we showed you earlier so those are all the documents that are provided in this it's much longer has a lot more detail we provide a lot more templates and if you have any questions don't hesitate to contact me thank you have a great evening
Show moreFrequently asked questions
How do I eSign a document before sending it?
How can I make an eSigned document expire?
How can I electronically sign a PDF document on my phone?
Get more for esign protocol made easy
- Signature block Kiosk Mode
- Prove electronically signed Non-Disclosure Agreement
- Endorse digi-sign Church Membership
- Authorize signature service Sales Report
- Anneal mark House Cleaning Proposal Template
- Justify esign Joint Marketing Agreement Template
- Try countersign Joinder Agreement
- Add Pledge Agreement initial
- Send Painting Proposal Template signature
- Fax Beach Party Invitation email signature
- Seal Music Camp Registration digital signature
- Password SEO Proposal Template electronically signed
- Pass Affidavit of Identity byline
- Renew General Bill of Sale esign
- Test Cleaning Work Order signature block
- Require Partnership Agreement Template signature service
- Comment beholder digi-sign
- Boost onlooker signed
- Compel cosigner mark
- Void Snow Removal Contract Template template electronically sign
- Adopt Usage Agreement template countersignature
- Vouch Video Production Quote template digital signature
- Establish College Room Agreement template signed
- Clear Hotel Business Plan Template template digi-sign
- Complete Camper Parental Authorization template esign
- Force Medical Power of Attorney Template template digital sign
- Permit Rental Deposit Receipt template initial
- Customize Affidavit of Service template signature