Draw Electronic signature Presentation Easy
Make the most out of your eSignature workflows with airSlate SignNow
Extensive suite of eSignature tools
Robust integration and API capabilities
Advanced security and compliance
Various collaboration tools
Enjoyable and stress-free signing experience
Extensive support
How Do I Save Electronic Signature in Word
Keep your eSignature workflows on track
Our user reviews speak for themselves






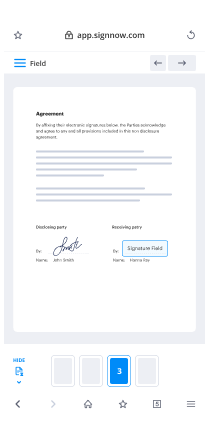
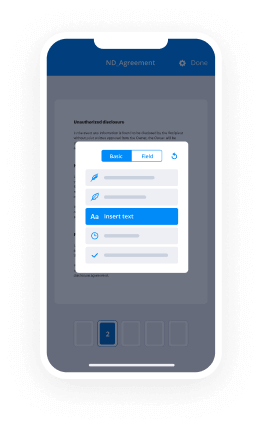
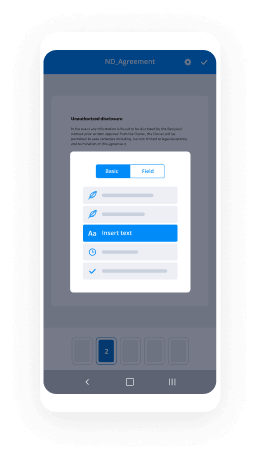
Draw Electronic signature Presentation Easy. Explore the most consumer-pleasant exposure to airSlate SignNow. Handle your entire file finalizing and discussing process electronically. Range from portable, pieces of paper-based and erroneous workflows to computerized, electronic digital and perfect. It is simple to make, provide and indication any papers on any device anyplace. Ensure that your important company situations don't fall overboard.
Find out how to Draw Electronic signature Presentation Easy. Keep to the easy manual to begin:
- Build your airSlate SignNow profile in mouse clicks or log on together with your Facebook or Google account.
- Take advantage of the 30-day time free trial version or pick a pricing plan that's great for you.
- Get any authorized template, build online fillable forms and reveal them tightly.
- Use sophisticated characteristics to Draw Electronic signature Presentation Easy.
- Sign, modify putting your signature on get and gather in-individual signatures ten times speedier.
- Establish auto reminders and receive notices at every phase.
Relocating your jobs into airSlate SignNow is uncomplicated. What practices is a straightforward method to Draw Electronic signature Presentation Easy, as well as suggestions and also hardwearing . fellow workers and associates for far better collaboration. Encourage your employees with all the very best instruments to stay on the top of business functions. Improve efficiency and level your company quicker.
How it works
Rate your experience
-
Best ROI. Our customers achieve an average 7x ROI within the first six months.
-
Scales with your use cases. From SMBs to mid-market, airSlate SignNow delivers results for businesses of all sizes.
-
Intuitive UI and API. Sign and send documents from your apps in minutes.
A smarter way to work: —how to industry sign banking integrate
FAQs
-
How was Balaji Viswanathan's overall experience attending the Global Entrepreneurship Summit 2017 held in Hyderabad, Where his I
Honestly, it was overwhelming. Things went truly viral and we were completely unprepared. We were so unprepared that we left the arena while there were important dignitaries trying to talk to us. Thanks to all of you for making this magical and thanks for your wonderful compliments.I was just a few feet from Mr.Modi as we entered the place and it was an amazing feeling. Someday I will get to talk with him. And Ivanka was probably the most beautiful woman I have met. We just joke that even our robot went giddy infront of these amazing personalities. Top entrepreneurs like Ritesh Aggarwal [founder of Oyo] walked up to us to congratulate and it was a magical feeling.The highlight of the whole thing was Amul making its signature billboard with our robot. I feel humbled by these billboards.We were truly excited by Shri Modi’s and Ms. Ivanka’s expression and the thousands of snaps that were taken along with hundreds of media coverage we got. The PM tweeted about it and his office featured the bot in their homepage.Yesterday in Finland at the Slush event a founder of a European company building robots walked up to me after recognizing the Mitra and talked about partnership in taking our robots to the European market. That felt elating [inspite of the fact that the product was not in the perfect shape especially after disassembling for the flight]. Many Finnish people took selfies with the robot to put it on their timeline and that was great.We also had a delegation from Japan’s Olympics committee visit our office to explore robots for the upcoming Tokyo Olympics. These sales might or might not happen in reality, but it felt amazing to imagine that Europe and Japan could buy products from India not for cost, but for uniqueness.To our well wishers:I’m humbled by your comments and it comes as a great boost for my team that has been tired and dazzled from months of hard work. Everyone in my team come from humble backgrounds and are out to prove something big and this comes as a shot in their arm.They worked day in and night to get the models going. We faced a number of setbacks. A model made in China never came home as it is stuck in Chennai customs for 2 months. A clay model we made had several inaccuracies and could never be completed. An android device we planned didn’t support our code base and we had to go for a quick fix with another tablet. Logistics went crazy and so did a million things.There were many hands that went in. Our CTO Bharath built the overall mechanics and everything to get it moving. He was helped by our production engineer Ram and Salman who helped in the structure. Our BDM Kaundinya pulled off this event and got our robot in after our COO Mahalakshmi was invited as a delegate for the event. Mahalakshmi and her brother Sudarshan did the whole supply chain and logistics for getting all the components. Her father helped us secure funding resources to sustain the R&D.Our electronics engineers Murali, Raushan and our new hire Spandana helped in building the circuit boards and making the electronics work. Our software engineers Ashwin, Anand and Sreejit made the entire stack. Our project manager Amit made sure the things went on schedule. and Our clay modeller Mr. Angappan helped us prepare the final fibreglass body. Our designers Vinay Rao and others at Bang Design helped us design the body. And journalists like Ranjani Ayyar and Nilesh Christopher spent time understanding the product to build an accurate story.And a special thanks to Kishlay Raj and his team at Sumeru. Their CEO and CTO gave their team’s personal time to finetune the underlying product. While we couldn’t showcase Hindi language NLP in the limited time it is a part of the product that we would sell.It took the whole village to put up this show.To the general public:Just to clarify, it was not any approval from the Indian government for being best startup or anything. And it didn’t involve me using any connections or favours. The event organisers were looking at something unique for the event. They were set on a large, humanoid robot to move around and greet the dignitaries and ours was the only one in the market - not just Indian - that could do so. We have done many events across India with our robots and we had worked with the event organisers before.A few days before the event, our robot was even cancelled for the event due to security reasons etc. Then we got back in the game once the senior leadership Wizcraft [the organisers] realised this was Made in India and they wanted to encourage such a product. And being a poor bootstrapped startup, they recognized the need to encourage entrepreneurship of all kinds. We got to this place without any angel/VC money.We have had many successes so far this year. Nasscom put us in a number of top events - including letting us pitch to the Japanese delegation at the Ministry of IT earlier this month. We were among the top 50 startups chosen by them and were in almost every top Nasscom event this year. We were also invited as a part of the Indian delegation to China and close to closing a big order there.We have also worked with Thub and had the honour of presenting a PoC to the global leadership of Novartis who came to Hyderabad earlier this year.To our critics:A few people on the Internet trolled us saying this looks like 70s toys, 90s toys, children’s project etc. I’m used to getting trolled and don’t mind criticism on me. But, since you guys were abusing the work of a group, I’m forced to respond in kind.I just ask them if it were that easy to make our product, why was there never a robot in a major Indian event so far? Why didn’t they make it and take all the honours? Why are you reading about an Indian robot doing such a thing for the first time? And why are there no global product companies from India, if making hardware is a child’s game? Think about it.People who never make things are usually the ones who underestimate the complexity that goes into a product. If you have never made things, you won’t know the pain of making.People said I pulled my connections. The event organizers don’t know anything about me and neither does anyone at BJP’s top brass or the US government that sponsored the event. Our event participation was pulled off by our excellent BDM and I had little to do with it. The event organizers would draw a blank if you ask them who Balaji is.Some people said I should learn from Elon Musk and Steve Jobs in how not to make shitty product. With due respect, I studied business history more than most of my critics. Jobs and Musk are geniuses. But even the geniuses start with more achievable targets. Elon Musk started with a product like this: Yellow Pages. Steve Jobs started with a product like this: Apple I. Honda’s Asimo started this way: Honda E1. Benz started this way: karl benz. Our own amazing ISRO guys started like this: The Story of How ISRO Defied All Odds To Put SatellitesGreat looking products don’t sprout in thin air. They are a result of years of work, starting with more modest looking creations. You cannot become an adult without starting as a fetus.We are trying to prove that there is a mass market for humanoids. It would be suicidal for a company of our stage to worry about a perfect product. Ours is more like a fetus and we are trying to keep it nourished and nurtured for a grand future.A few more critics said we should have been embarrassed to put such a product. Predictably none of those are entrepreneurs or someone who have created something. Because, real creators and entrepreneurs don’t worry about breaking things. Taking risk is what we do. Rather than an embarrassment, I would feel elated for my team even if it had not even started moving.The challenges:Try moving a 50kg, 5 feet tall object through varying terrain in a noiseless, smooth way without tripping and you will understand the physics behind the design. And with a battery life to last hours of rehearsals. It is a non-trivial piece of engineering with suspension systems that my team created. From the fiberglass body to the internal structure, everything was made here.We built the hardware, software and electronics for this. We built 9 prototypes this year. We are bootstrapped and have done all this hardware investment without any external funding. And while we didn’t show its real capabilities in the GES event [too risky to try] our robots can move autonomously and that what the funky head antenna is for. We integrated NLP tools and face recognition APIs and those are non-trivial things. And over the years we iterated a million times to get things better and better.Of course, there are plenty of inaccuracies and faults. This was handmade and in a very short span of time. But, the team has gone through enormous things to get this far, from where we started earlier this year.Developing product in India is hard. Making everything in India is hard. Making such a large object is hard as the mould costs multiply and the complexity goes as a square of that. Talk is easy, getting the stuff work on the floor is hard and why these don’t happen often.I’m not saying that our product is perfect. I’m saying the product is perfected and we will get there someday. If we keep worrying about breaking things, we are not a startup.In conclusion:I’m not saying we are India’s best product or even India’s best robotics company. The GES event was not about that. They wanted to showcase entrepreneurship at an early stage and a product that fit their needs. Large finished products come from large companies, startups backed by billionaires or at least startups with large venture funding. GES thought having a product from a company that didn’t fit any of these categories above and supported by no one was worth doing.Hardware in India will sprout only when difficulties of building hardware is understood and its complexities appreciated. Everyone says they or their grandmother or their child could have our product. But, they didn’t. Only our team did. And it is much easy imagining doing a product than doing it.Is the product overhyped? Maybe. Was there luck involved. Yes. But hey for a bootstrapped startup that has been teetering on brink of bankruptcy and has had more than its share of misfortune this year, I would not mind a little luck. Would you? Peace!
-
How does bitcoin work? Who actually pays for the mining done?
Approximately once every 10 minutes since January 3, 2009 at 18:15:05 UTC, one miner in the world has found/will find a valid Bitcoin block that meets the current difficulty requirements. When that happens, the block reward and the transaction fees will be distributed to the wallet(s) configured by that miner in that block. These two sources make up all of a miner’s income.Block rewards are the only way that new Bitcoins are min[t]ed. The block reward started at 50 BTC/block and halves every 210,000 blocks. This is why the current block reward is 12.5 BTC/block as of Sept 2017.Transaction fees for a block are the sum of the fees paid for all new transactions included in that block. For example, I might send you .5 BTC and specify a fee of .00004 BTC as incentive for miners to include that transaction in their block. The first miner to include that transaction in an accepted block will then earn that .00004 BTC in addition to the block reward and fees from other included transactions.Early on, transaction fees were a negligible source of miner income since there were very few transactions and the block reward was high. However, as the block reward diminishes and the number of transactions grows this trend will reverse and block rewards will become negligible and transaction fees will dominate earnings.Mining pools are another layer on top of this. Instead of the reward and fees going to a single miner, they instead have a number of miners pooling their efforts and splitting the rewards based on that pool's rules. In this way, if it would normally take you 10,000 years on average to find a block by yourself, you can instead join a mining pool and get small fractions of a block reward regularly based on your mining contribution and the pool's rules.Compare that to solo mining without a mining pool, where until you find a valid block you won't get a single Satoshi.
-
Are all the other universes connected to each other? If yes, how?
Cosmic Variance : Everything is ConnectedThey do things differently over in Britain. For one thing, their idea of a fun and entertaining night out includes going to listen to a lecture/demonstration on quantum mechanics and the laws of physics. Of course, it helps when the lecture is given by someone as charismatic as Brian Cox, and the front row seats are filled with celebrities. (And yes I know, there are people here in the US who would find that entertaining as well — I’m one of them.) In particular, this snippet about harmonics and QM has gotten a lot of well-deserved play on the intertubes.More recently, though, another excerpt from this lecture has been passed around, this one about ramifications of the Pauli Exclusion Principle. (Headline at io9: “Brian Cox explains the interconnectedness of the universe, explodes your brain.”)The problem is that, in this video, the proffered mind-bending consequences of quantum mechanics aren’t actually correct. Some people pointed this out, including Tom Swanson in a somewhat intemperately-worded blog post, to which I pointed in a tweet. Which led to some tiresome sniping on Twitter, which you can dig up if you’re really fascinated. Much more interesting to me is getting the physics right.One thing should be clear: getting the physics right isn’t easy. For one thing, going from simple quantum problems of a single particle in a textbook to the messy real world is often a complicated and confusing process. For another, the measurement process in quantum mechanics is famously confusing and not completely settled, even among professional physicists.And finally, when one translates from the relative clarity of the equations to a natural-language description in order to signNow a broad audience, it’s always possible to quibble about the best way to translate. It’s completely unfair in these situations to declare a certain popular exposition “wrong” just because it isn’t the way you would have done it, or even because it assumes certain technical details that the presenter did not fully footnote. It’s a popular lecture, not a scholarly tome. In this kind of format, there are two relevant questions: (1) is there an interpretation of what’s being said that matches the informal description onto a correct formal statement within the mathematical formulation of the theory?; and (2) has the formalism been translated in such a way that a non-expert listener will come away with an understanding that is reasonably close to reality? We should be charitable interpreters, in other words.In the video, Cox displays a piece of diamond, in order to illustrate the Pauli Exclusion Principle. The exclusion principle says that no two fermions — “matter” particles in quantum mechanics, as contrasted with the boson “force” particles — can exist in exactly the same quantum state. This principle is why chemistry is interesting, because electrons have to have increasingly baroque-looking orbitals in order to be bound to the same atom. It’s also why matter (like diamond) is solid, because atoms can’t all be squeezed into the same place. So far, so good.But then he tries to draw a more profound conclusion: that interacting with the diamond right here instantaneously affects every electron in the universe. Here’s the quote:So here’s the amazing thing: the exclusion principle still applies, so none of the electrons in the universe can sit in precisely the same energy level. But that must mean something very odd. See, let me take this diamond, and let me just heat it up a bit between my hands. Just gently warming it up, and put a bit of energy into it, so I’m shifting the electrons around. Some of the electrons are jumping into different energy levels. But this shift of the electron configuration inside the diamond has consequences, because the sum total of all the electrons in the universe must respect Pauli. Therefore, every electron around every atom in the universe must be shifted as I heat the diamond up to make sure that none of them end up in the same energy level. When I heat this diamond up all the electrons across the universe instantly but imperceptibly change their energy levels.(Minor quibble: I don’t think that rubbing the diamond causes any “jumping” of electrons; the heating comes from exciting vibrational modes of the atoms in the crystal. But maybe I’m wrong about that? And in any event it’s irrelevant to this particular discussion.)At face value, there’s no question that what he says here lies somewhere between misleading and wrong. It seems quite plain (that’s the problem with being a clear speaker) that he’s saying that the energy levels of electrons throughout the universe must change because we’ve changed the energy levels of some electrons here in the diamond, and the Pauli exclusion principle says that two electrons can’t be in the same energy level. But the exclusion principle doesn’t say that; it says that no two identical particles can be in the same quantum state. The energy is part of a quantum state, but doesn’t define it completely; we need to include other things like the position, or the spin. (The ground state of a helium atom, for example, has two electrons with precisely the same energy, just different spins.)Consider a box with non-interacting fermions, all in distinct quantum states (as they must be). Take just one of them and zap it to move it into a different quantum state, one unoccupied by any other particle. What happens to the other particles in the box? Precisely nothing. Of course if you zap it into a quantum state that is already occupied by another particle, that particle gets bumped somewhere else — but in the real universe there are vastly more unoccupied states than occupied ones, so that can’t be what’s going on. Taken literally as a consequence of the exclusion principle, the statement is wrong.But it’s possible that there is a more carefully-worded version of the statement that relies on other physics and is correct. And we might learn some physics by thinking about it, so it’s worth a bit of effort. I think it’s possible to come up with interpretations of the statement that make it correct, but in doing so the implications become so completely different from what the audience actually heard that I don’t think we can give it a pass.The two possibilities for additional physics (over and above the exclusion principle) that could be taken into account to make the statement true are (1) electromagnetic interactions of the electrons, and (2) quantum entanglement and collapse of the wave function. Let’s look at each in turn.The first possibility, and the one I actually think is lurking behind Cox’s explanation, is that electrons aren’t simply non-interacting fermions; they have an electric field, which means they can interact with other electrons, not to mention protons and other charged particles. If we change the ambient electric field — e.g., by moving the diamond around — it changes the wave function of the electrons, because the energy changes. Physicists would say the we changed the Hamiltonian, the expression for the energy of the system.There is an interesting and important point to be made here: in quantum mechanics, the wave function for a particle will generically be spread out all over the universe, not confined to a small region. In practice, the overwhelming majority of the wave function might be localized to one particular place, but in principle there’s a very tiny bit of it at almost every point in space. (At some points it might be precisely zero, but those will be relatively rare.) Consequently, when I change the electric field anywhere in the universe, in principle the wave function of every electron changes just a little bit. I suspect that is the physical effect that Cox is relying on in his explanation.But there are serious problems in accepting this as an interpretation of what he actually said. For one thing, it has nothing to do with the exclusion principle; bosons (who can happily pile on top of each other in the same quantum state) would be affected just as much as fermions. More importantly, it fails as a job of translation, by giving people a completely incorrect idea of what is going on.The point of this last statement is that when you say “When I heat this diamond up all the electrons across the universe instantly but imperceptibly change their energy levels,” people are naturally going to believe that something has changed about electrons very far away. But that’s not true, in the most accurate meaning we can attach to those words. In particular, imagine there is some physicist located in the Andromeda galaxy, doing experiments on the energy levels of electrons. This is a really good experimenter, with lots of electrons available and the ability to measure energies to arbitrarily good precision. When we rub the diamond here on Earth, is there any change at all in what that experimenter would measure?Of course the answer is “none whatsoever.” Not just in practice, but in principle. The Hamiltonian of the universe will change when we heat up the diamond, which changes the instantaneous time-independent solutions to the Schoedinger equation throughout space, so in principle the energy levels of all the electrons in the universe do change. But that change is completely invisible to the far-off experimenter; there will be a change, but it won’t happen until the change in the electromagnetic field itself has had time to propagate out to Andromeda, which is at the speed of light. Another way of saying it is that “energy levels” are static, unchanging states, and what really happens is that we poke the electron into a non-static state that gradually evolves. (If it were any other way, we could send signals faster than light using this technique.)Verdict: if this is what’s going on, there is an interpretation under which Cox’s statement is correct, except that it has nothing to do with the exclusion principle, and more importantly it gives a quite false impression to anyone who might be listening.The other possibly relevant bit of physics is quantum entanglement and wave function collapse. This is usually the topic where people start talking about instantaneous changes throughout space, and we get mired in interpretive messes. Again, these concepts weren’t mentioned in this part of the lecture, and aren’t directly tied to the exclusion principle, but it’s worth discussing them.There is something amazing and magical about quantum mechanics that is worth emphasizing over and over again. To wit: unlike in classical mechanics, there are not separate states for every particle in the universe. There is only one state, describing all the particles; modest people call it the “many-particle wave function,” while visionaries call it the “wave function of the universe.” But the point is that you can’t necessarily describe (or measure) what one particle is doing without also having implications for what other particles are doing — even “instantaneously” throughout space (although in ways that have to be carefully parsed).Imagine we have a situation with two electrons, each in a separate atom, with different energy levels in each atom. Quantum mechanics tells us that it’s possible for the system to be in the following kind of state: each electron is either in energy level 1 or energy level 2, and we don’t know which one (more carefully, they are in a superposition), but we do know that they are in different energy levels. So if we measure the first electron and find it in level 1, we know for sure that the other electron is in level 2, and vice-versa. This is true even if the two electrons are a jillion miles away from each other.As far as I can tell, this isn’t at all what Brian Cox was talking about; he discusses heating up the electrons in a diamond by rubbing on it, not measuring their energies by observing them and then drawing conclusions about entangled electrons very far away. (In a real-world context it’s very unlikely that distant electrons are entangled in any noticeable way, although strictly speaking you could argue that everything is slightly entangled with everything else.) But there is some underlying moral similarity — this is, as mentioned, the context in which people traditionally talk about instantaneous changed in quantum mechanics.So let’s go back to our observer in Andromeda. Imagine that we have such a situation with two electrons in two atoms, in a mutually entangled state. We measure our electron to be in energy level 1. Is it true that we instantly know that our far-away friend will measure their electron to be in energy level 2? Yes, absolutely true.But consider the same experiment from the point of view of our far-away friend. They know what the state of the electrons is, so they know that when they observe their electron it will be either in level 1 or level 2, and ours will be in the other one. And let’s say they even know that we are going to make a measurement at some particular moment in time. What changes about any measurement they could make on their electron, before and after we measure ours?Absolutely nothing. Before we made our measurement, they didn’t know the energy level of their electron, and would give 50/50 chances for finding it in level 1 or 2. After we made our measurement, it’s in some particular state, but they don’t know what that state is. So again they would give a 50/50 chance for getting either result. From their point of view, nothing has changed.It has to work out this way, of course. Otherwise we could indeed use quantum entanglement to send signals faster than light (which we can’t). Indeed, note that we had to refer to “time” in some particular reference frame, stretching across millions of light-years. In some other frame, relativity teaches us that the order of measurements could be completely different. So it can’t actually matter. It’s possible to say that the wave function of the universe changes instantaneously throughout space when we make a measurement; but that statement has no consequences. It’s just one of an infinite number of legitimate descriptions of the situation, corresponding to different choices of how we define “time.”Verdict: I don’t think this is what Cox was talking about. He doesn’t mention entanglement, or collapse of the wave function, or anything like that. But even if he had, I would personally judge it extremely misleading to tell people that the energy of very far-away electrons suddenly changed because I was rubbing a diamond here in this room.Just to complicate things a bit more, Brian in a tweet refers to this discussion of the double-well potential as some quantitative justification for what he’s getting at in the lecture. These notes are a bit confusing, but I’ve had a go at them.The reason they are confusing is because they start off talking about the exclusion principle and indistinguishable particles, but when it comes time to look at equations they only consider single-particle quantum mechanics. They have a situation with two “potential wells” — think of two atoms, perhaps quite far away, in which an electron might find itself. They then consider the wave function for a single electron, ψ(x). And they show, perfectly correctly, that the lowest energy states of this system have nearly identical energies, and have the feature that the electron has an equal probability of being in either of the two atoms.Which, as far as it goes, is completely fine. It illustrates an interesting example where the lowest-energy state of the electron can be really spread out in space, rather than being localized on a single atom. In particular, the very existence of the other atom far away has a tiny but (in principle) perceptible effect on the shape of the wave function in the vicinity of the nearby atom.But this says very little about what we purportedly care about, which is the Pauli exclusion principle, something that only makes sense when we have more than one electron. (It says that no two electrons can be in the same state; it has nothing interesting to say about what one electron can do.) It’s almost as if the notes cut off before they could be finished. If we wanted to think about the exclusion principle, we would need to think about two electrons, with positions let’s say x1 and x2, and a joint quantum wave function ψ(x1, x2). Then we would note that fermions have the property that such a wave function must be “odd” in its arguments: ψ(x1, x2) = -ψ(x2, x1). Physically, we’re saying that the wave function goes to minus itself when we exchange the two particles. But if the two particles were in exactly the same state, the wave function would necessarily be unchanged when we exchanged the particles. And a function that is both equal to another function and equal to minus that function is necessarily zero. So that’s the exclusion principle: given that minus sign under exchange, two particles can never be in precisely the same quantum state.The notes don’t say any of that, however; they just talk about the two lowest energy levels in a double-well potential for a single electron. They don’t demonstrate anything interesting about the exclusion principle. The analysis does imply, correctly, that changing the Hamiltonian of a particle somewhere far away (e.g. by altering the shape of one of the wells) changes, even if by just a little bit, the energy of the wave function defined over all space. That’s connected to the first possible interpretation of Cox’s lecture above, that heating up the diamond changes the Hamiltonian of the universe and therefore affects the wave function of every electron. Which also has nothing to do with the exclusion principle, so at least it’s consistent.In terms of explaining the mysteries of quantum mechanics to a wide audience, which is the point here, I think the bottom line is this: rubbing a diamond here in this room does not have any instantaneous effect whatsoever on experiments being done on electrons very far away. There are two very interesting and conceptually central points worth making: that the Pauli exclusion principle helps explain the stability of matter, and that quantum mechanics says there is a single state for the whole universe rather than separate states for each individual particle. But in this case these became mixed up a bit, and I suspect that this part of the lecture wasn’t the most edifying for the audience. (The rest of the lecture still remains pretty awesome.)Update: I added this as a comment, but I’m promoting it to the body of the post because hopefully it makes things clearer for people who like a bit more technical precision in their quantum mechanics. [Note the mid-update extra update.]Consider the double-well potential talked about in the notes I linked to near the end of the post. Think of this as representing two hydrogen nuclei, very far away. And imagine two electrons in this background, close to their ground states.To start, think of the electrons as free particles, not interacting with each other. (That’s a very bad approximation in this case, contrary to what is said in the notes, but we can fix it later.) As the notes correctly state, for any single electron there will be two low-lying states, one that is even E(x) and one that is odd O(x). When we now add the other electron in, they can’t both be in the same lowest-lying state (the even one), because that would violate Pauli. So you are tempted to put one in E(x1) and the other in O(2).But that’s not right, because they’re indistinguishable fermions. The two-particle wave function needs to obey ψ(x1, x2) = -ψ(x2, x1). So the correct state is the antisymmetric product: ψ(x1, x2) = E(x1) O(x2) – O(x1) E(x2).That means that neither electron is really in an energy level; they are both part of an entangled superposition. If you zap one of them into a completely different energy, nothing whatsoever happens to the other one. It would now be possible for the other one to decay to be purely in the ground state, rather than a superposition of E and O, but that would require some interaction to allow the decay. (All this is ignoring spins. If we allow for spin, they could both be in the ground-state energy level, just with opposite spins. When we zapped one, what happens to the other is again precisely nothing. That’s what you get for considering non-interacting particles.)[Second update: the below two italicized paragraphs are wrong, my bad. It’s actually quite a good approximation (although still an approximation) to ignore the electromagnetic interactions of the electrons, because after antisymmetrization you will almost always find precisely one electron in each well. If electrons were bosons, you’d get a similar quantum state because the interactions would be important, but for fermions the exclusion principle does the job. Final paragraph is still okay.]But of course it’s a very bad approximation to ignore the interaction between the two electrons, precisely because of the above analysis; it’s not true that one is here and one is far away, they both are equally distributed between being here and being far away, and can interact noticeably.Since electrons repel, the true ground state is one in which the wave function for one is strongly concentrated one one hydrogen atom, and the wave function for the other is strongly concentrated on the other. Of course it’s the antisymmetrized product of those two possibilities, because they are identical fermions. The energies of both are identical.Now when you zap one electron to change its energy, you do change the energy of the other one, in principle. But it has nothing to do with the exclusion principle; it’s just because you’ve changed the amount of electrostatic repulsion by changing the spatial wave function of one of the electrons.Furthermore, while you instantaneously change “the energy levels” available to the far-away electron by jiggling the one nearby, you don’t actually change the position-space wave function in the far-away region at all. As I said in the post, you’ve poked the other electron into a superposition rather than being in an energy eigenstate. Its wave function (to the extent that we can talk about it, e.g. by integrating out the other particles) is now a function of time. And the place where it’s actually evolving is completely inside your light cone, not infinitely far away. So there is literally nothing someone could do, in principle as well as practice, to detect any change as a far-away observer.Scientists find first evidence that many universes existThe signatures of a bubble collision: A collision (top left) induces a temperature modulation in the CMB temperature map (top right). The “blob” associated with the collision is identified by a large needlet response (bottom left).By looking far out into space and observing what’s going on there, scientists have been led to theorize that it all started with a Big Bang, immediately followed by a brief period of super-accelerated expansion called inflation. Perhaps this was the beginning of everything, but lately a few scientists have been wondering if something could have come before that, setting up the initial conditions for the birth of our universe.In the most recent study on pre-Big Bang science posted at arXiv.org e-Print archive, a team of researchers from the UK, Canada, and the US, Stephen M. Feeney, et al, have revealed that they have discovered four statistically unlikely circular patterns in the cosmic microwave background (CMB). The researchers think that these marks could be “bruises” that our universe has incurred from being bumped four times by other universes. If they turn out to be correct, it would be the first evidence that universes other than ours do exist.The idea that there are many other universes out there is not new, as scientists have previously suggested that we live in a “multiverse” consisting of an infinite number of universes. The multiverse concept stems from the idea of eternal inflation, in which the inflationary period that our universe went through right after the Big Bang was just one of many inflationary periods that different parts of space were and are still undergoing. When one part of space undergoes one of these dramatic growth spurts, it balloons into its own universe with its own physical properties. As its name suggests, eternal inflation occurs an infinite number of times, creating an infinite number of universes, resulting in the multiverse.These infinite universes are sometimes called bubble universes even though they are irregular-shaped, not round. The bubble universes can move around and occasionally collide with other bubble universes. As Feeney, et al., explain in their paper, these collisions produce inhomogeneities in the inner-bubble cosmology, which could appear in the CMB. The scientists developed an algorithm to search for bubble collisions in the CMB with specific properties, which led them to find the four circular patterns.Still, the scientists acknowledge that it is rather easy to find a variety of statistically unlikely properties in a large dataset like the CMB. The researchers emphasize that more work is needed to confirm this claim, which could come in short time from the Planck satellite, which has a resolution three times better than that of WMAP (where the current data comes from), as well as an order of magnitude greater sensitivity. Nevertheless, they hope that the search for bubble collisions could provide some insight into the history of our universe, whether or not the collisions turn out to be real.“The conclusive non-detection of a bubble collision can be used to place stringent limits on theories giving rise to eternal inflation; however, if a bubble collision is verified by future data, then we will gain an insight not only into our own universebut a multiverse beyond,” the researchers write in their study.This is the second study in the past month that has used CMB data to search for what could have occurred before the Big Bang. In the first study, Roger Penrose and Vahe Gurzadyan found concentric circles with lower-than-average temperature variation in the CMB, which could be evidence for a cyclic cosmology in which Big Bangs occur over and over.
-
Will it be viable one day to mine the Moon and extract the resources there for something useful?
Yes it is feasible. There’s a long way from it being feasible in principle and an actual business, but plenty of possibilities to explore.The main suggestions include volatiles from the poles - supplying water and the water split into hydrogen and oxygen as fuel, to LEO - where the Moon has the advantage that export is much easier than from Earth, precious metals for export to Earth such as platinum, which may be there as a result of impacts of iron rich meteorites and giant asteroids, and many resources suggested that could be used in situ on the Moon. We could also create solar panels on the Moon. It’s useful for fabricating electronics because of the hard vacuum. There are some processes you can do on the Moon easily which would be hard to do on Earth because it is so difficult to get a sufficiently hard vacuum to do them.There are several books by Moon enthusiasts describing this in detail, how it would work. Paul Spudis is one, with his most recent book, The Value of the Moon: How to Explore, Live, and Prosper in Space Using the Moon's Resources. Another is Dennis Wingo, CEO of Skycorp, and author of Moonrush, see his recent paper, and appearance on the Space Show. Others include Madhu Thangavelu, David Schrunk, and other authors and contributors to The Moon: Resources, Future Development and Settlement. See also David Schrunk's paper Planet Moon Philosophy , and their appearance on The Space Show.I did a summary of some of the main resources on the Moon for my Case for Moon first. The rest of this answer consists of extracts from the section The Moon is resource rich from my kindle book.VOLATILE RESOURCESWe have pretty good evidence now of ice at the poles, in permanently shadowed craters, thought to be relatively pure and at least a couple of meters thick according to radar data from a NASA instrument flying on India's Chandrayaan-1 lunar orbiter.It's not a direct detection however, so there is still room for scepticism about it, as rough material would have the same radar signature as radar transparent ice. But craters that are rough when new, are rough both inside and outside the crater rim. While these signatures are found only inside the craters and not outside the rims, which they interpret as meaning that they are caused by ice. The temperatures are also right for ice.If it is ice, it could be "fluffy ice"."We do not know the physical characteristics of this ice—solid, dense ice, or “fairy castle”—snow-like ice would have similar radar properties. In possible support of the latter, the low radar albedo and lower than typical CPR values for nonanomalous terrain near the polar craters are 0.2–0.3, somewhat lower than normal for the nonpolar highlands terrain of the Moon and are suggesting the presence of a low density, “fluffy” surface."(page 13 of Evidence for water ice on the moon: Results for anomalous polar)In either case, it is not just a little ice; if this is what they detected, there's estimated to be at least 600 million metric tons of this, and possibly much more.It also contains other volatiles. We know for sure that there is some ice on the Moon, by the LCROSS impact experiment. Relative to H2O at 100% they found H2S at 16.75%, NH3 at 6.03% SO2 at 3.19%, C2H4 at 3.12%, CO2 at 2.17%.So, if the rest of the ice at the poles has a similar constitution to the impact site that's a lot of nitrogen (in the ammonia) and CO2on the Moon at the poles.On the other hand, caution is needed as this is not direct detection. The LEND results (searching for hydrogen through reduced emissions of neutrons of a particular type) are particularly puzzling, as there is almost no resemblance between their map and the miniSAR map.LEND map - in this picture blue is reduced neutron emission and shows likely locations of hydrogen. 0 degrees longitude is at the top.They did detect hydrogen, but puzzlingly, it was not correlated with the permanently shadowed regions - there was some hydrogen in permanently shadowed regions, and some also in illuminated regions. A recent paper suggests that ice mixed in the regolith in illuminated regions may be ancient ice that survived a minor shift of the lunar axis.According to one hypothesis, this may be ancient deposits from over three billion years ago before volcanic activity, which changed the polar axis slightly by shifting material.A new LEND mission has been proposed involving low passes over the poles at altitudes as low as a few kilometers, for higher resolution results.The Moon may also have ice at lower latitudes too, as there are permanently shaded regions up to 58 degrees from the poles (only 32 degrees from the equator). Though these regions are too warm to have ice on the surface, there may be ice there underground. See Ice may lurk in shadows beyond Moon's poles (Nature, 2012).At any rate, the Moon does seem to have resources of ice at the poles (though memorably, Patrick Moore in one of the last Sky at Night programs that he did said that he'd believe there is ice at the poles when someone brought him a glass of water from the Moon). More research is needed to find out how much there is and where it is.METALSCritics often say that the Moon is undifferentiated and doesn't have any processes to concentrate ores. Although the Moon doesn't have any liquid water so all the processes involving concentration of resources through water erosion won't work, it still has many processes that can concentrate ores. Including:Fractional crystallization - as a melt cools down, some minerals crystallize out at a higher temperature than others so form first. They then settle or float, so remove the chemical components that make them up from the mix, so changing its formula, leading to new crystals to form in a sequence.Gravitational settling, lower mass material floats to the top.Volcanic outgassing can concentrate materials such as iron, sulfur, chlorine, zinc, cadmium, gold, silver and lead.The processes that lead to volatiles condensing at the poles - which it seems can also concentrate silver tooProcesses unique to the Moon (perhaps electrostatic dust levitation may concentrate materials)?Volatiles brought in as part of the solar windAsteroid and micrometeorite impacts bring materials from asteroids to the lunar surface such as iron and possibly platinum group metals etc.The Moon has many valuable ores for metals. For instance, the highland regions (probably the original crust of th Moon) consists mainly of Anorthite (a form of feldspar, formula CaAl2Si2O8) which is 20% Aluminium, compared with 25% Aluminium for Bauxite on Earth. So aluminium ores are abundant on the Moon, indeed orders of magnitude more abundant than they are in typical asteroids, but it does require a lot of energy to extract the aluminium from the ore. Either a nuclear power plant or large areas of solar panels. Crawford, in his "Lunar Resources: a Review", says this about aluminium on the Moon:"Aluminium (Al) is another potentially useful metal, with a concentration in lunar highland regoliths (typically10-18 wt%) that is orders of magnitude higher than occurs in likely asteroidal sources (i.e. ~1 wt% in carbonaceous and ordinary chondtites, and <0.01 wt% in iron meteorites; . It follows that, as for Ti, the Moon may become the preferred source for Al in cis-lunar space. Extraction of Al will require breaking down anorthitic plagioclase (CaAl2Si2O8), which is ubiquitous in the lunar highlands, but this will be energy intensive (e.g. via magma electrolysis or carbothermal reduction; Alternative, possibly less energy intensive, processes include the fluoridation process proposed by Landis , acid digestion of regolith to produce pure oxides followed by reduction of Al2O3 (Duke et al.), or a variant of the molten salt electrochemical process described by Schwandt et al."Mining this for the aluminium would create calcium as a byproduct, which is useful as a conductor in vacuum conditions, a better conductor than copper weight for weight -you need half the mass for the same amount of electricity. (Copper does better than calcium on a per volume basis because it is 5.8 times denser, it is also of course much more practical in an atmosphere because calcium reacts vigorously with air, but that's not a problem for conductors that operate in a lunar vacuum, and in space applications the reduced mass may be an advantage)."Calcium metal is not used as a conductor on Earth simply because calcium burns spontaneously when it comes in contact with oxygen (much like the pure magnesium metal in camera flashbulbs). But in vacuum environments in space, calcium becomes attractive."Calcium is a better electrical conductor than both aluminum and copper. Calcium's conductivity also holds up better against heating. A couple of figures mining engineer David Kuck pulled out of the scientific literature: "At [20C, 68F], calcium will conduct 16.7% more electricity than aluminum, and at [100C, 212F] it will conduct 21.6% more electricity through one centimeter length and one gram mass of the respective metal." Compared to copper, calcium will conduct two and a half times as much electricity at 20C, 68F, and 297% as much at 100C, 212F."Like copper, calcium metal is easy to work with. It is easily shaped and molded, machined, extruded into wire, pressed, and hammered."As would be expected of a highland element, calcium is lightweight, roughly half the density of aluminum. However, calcium is not a good construction material because it is not strong. Calcium also sublimes (evaporates) slowly in vacuum, so it may be necessary to coat calcium parts to prevent the calcium from slowly coating other important surfaces like mirrors. In fact, calcium is sometimes used to deoxidize some metal surfaces. Calcium doesn't melt until 845C (1553F)."Utilization of lunar materials will see the introduction of industrial applications of calcium metal in space."From the section on Mining the Moon in Permanent - by Mark Evan Prado, a physicist in the Washington, D.C., region working for the Pentagon in advanced planning in the space program.The Moon is deficient in copper, at least on the basis of what is known so far, but as well as calcium, aluminium is a good conductor.The LCROSS experiment found silver (a superb conductor) and mercury at the impact site, but the concentration is not known, except that it is far higher than the levels in the Apollo samples, and is probably in a layer below the surface, as the signal was delayed. See LCROSS mission may have struck silver on the moon.It has abundant iron - in addition to ores (which would need a lot of power to extract), it actually has free iron metalFrom meteorite impactsNanometer sized "blebs" released from the rock by the hydrogen in the solar wind reacting with iron oxidesParticles of iron concentrated from the source materials for the regolith.It's in powder form already, and naturally alloyed with nickel and cobalt. The blebs, or "nanophase iron" are found inside impact glass particles, so would be hard to extract. The rest though is made up of tiny particles of pure iron, so the obvious thing to try to do is to separate them out using powerful magnets. They are rather small though, most are less than a micron in diameter which could be a challenge. If we can separate them out, we can get five kilograms of iron, 300 grams of nickel and about half a gram of platinum, gold etc. (platinum group metals) in every cubic meter of regolith - as pure metal what's more. (This summarizes part of section 5, Metals from Crawford)He bases that on a paper from 1980 by Morris and particularly its conclusion, which uses a model to interpret the data. Taylor and Meeks in the section Agglutinitic Glass versus Grain Size and Maturity (page 133) in their paper suggest that perhaps most of the iron is in nanophase form, mixed up with the glass and hard to extract.However we don't need to speculate any more as Jayashree Sridhar et al of the NASA Johnson Space Center have done the experiment using actual samples of lunar regolith. See Extraction of meteoritic metals from lunar regolith, and they succeeded! The nanophase iron was a problem but they were able to work around it by varying the experimental setup. By varying on the size of particle they ground it down to, the strength of the magnets and details of the technique they could extract over 80% of the meteoritic iron in some of the tests. They conclude:"Experimental results indicate promise for the extraction of meteoritic metals from lunar regolith. However, more work is needed to refine the technique and understand more about the variables that affected our results."The iron is valuable for steel, and is also a conductor, though not nearly as good as Aluminium or Calcium. It would be useful for some applications such as electric railroads on Mars, and is a conductor easy to access in the early stages.Also nickel and iron are useful for making nickel / iron batteries. These could be useful for making batteries on the Moon with in situ resources, for instance to help last through the lunar night."Iron-nickel batteries are very rugged. Their lifetimes which can exceed 20 years are not affected by heat, cold or deep cycling. They are not easily damaged by rapid discharging or over-charging. On the downside, they have poor performance at low temperatures but they can be kept warm with insulation (e.g. simple regolith) and thermal wadis. Also, they only have a charge to discharge efficiency of 65% and will self discharge at the rate of 20% to 40% per month. Despite these shortcomings, they might be the Moon-made power storage systems of choice due to their simplicity and the availability of their component materials on the Moon. Moreover, these materials are among the easiest of materials to produce on the Moon."See Electrical Energy Storage Using Only Lunar Materials.Then, you also have titanium. This is especially interesting as it is rare in asteroids. Apollo 17 samples are 20% high purity Ilmenite, a Titanium ore which is found in the lunar mare. And better than that, the Lunar Reconnaissance Orbiter, with its spectral mapping of the Moon, discovered deposits that are up to 10% titanium, more than ten times higher than titanium ores on Earth. (Phys.org report, NASA image). Titanium is an industrially desirable metal, stronger per unit weight than Aluminium (though it is a poor conductor).Titanium is also widely used in medicine for hip replacements, dental implants, etc., as "one of the few metals human bone can grow around firmly", see also this new titanium / gold alloy four times tougher than titaniumTitanium is especially useful for medical applications because itForms an inert and stable titanium oxide layer spontaneouslyHas a high strength to weight ratioDoesn't leach into blood and other aqueous environments because of its low rate of ion formationIs one of the few materials that can integrate directly into living bone tissues (osseointegration) without any soft tissue layers in betweenCrawford writes (page 17):"Therefore, in the context of a future space economy, the Moon may have a signNow advantage over asteroids as a source of Ti. The fact that oxygen is also produced as a result of Ti production from ilmenite could make combined Ti/O2 production one of the more economically attractive future industries on the Moon.For more on this, see major lunar minerals. And for an in depth study, read Crawford's review.So, yes, there are plenty of metals on the Moon, but it might take a lot of power to extract them, apart from the iron, if that can be separated out using magnets.And that's mainly based on the Apollo results which explored a small region of the lunar surface which has been found to be in some ways unrepresentative. The Moon may have many other surprises in store. Many ores on Earth would not be detected from orbit, and it seems the Moon has a fairly complex geology as well.As an example of one way the Moon could surprise us - Earth is often hit by iron meteorites, so the Moon should be also. The main question is, how Dennis Wingo has hypothesized in his Moonrush book, that the Moon may also have valuable platinum group metals which could be mined, the result of the impacts of these iron meteorites.Taking this further, there's a hypothesis by Wieczorek et al that magnetic anomalies on the Moon around the south pole Aitken basin may be from the remains of the metal core of a large 110 km diameter differentiated asteroid that hit the Moon to form the basin. If so, they could be useful sources for platinum, gold, etc.From Wieczorek et al, the North and South poles are marked N and S. Notice the magnetic anomalies clustered around part of the rim of the South Pole Aitken Basin. This is thought to be the result of an impact by a 110 km diameter asteroid. Wieczorek et al hypothesize that the magnetic anomalies trace out the remains of the metal core of this asteroid. If so these could be rich ores, including iron, nickel, also platinum and other platinum group metals (gold, rhodium etc). See page 16 of Crawford's Lunar Resources: A ReviewPlatinum is a particularly useful metal. It is heavy, soft, malleable as gold and silver, easy to draw into wires, very unreactive, and has a high melting point. Out of gold, silver, platinum and copper, platinum is the densest and the hardest and the least reactive (the others are somewhat better in terms of electrical and thermal conductivity, and malleability, but it's not too bad at those either). So, it's not just useful for catalytic converters, fuel cells, dental fillings and jewelry. We'd probably use it a fair bit in other ways too if it didn't cost so much.The platinum group metals might be valuable enough to return to Earth from the Moon, just as suggested for the asteroids, especially if there is water to split and use as fuel available on the Moon or once they set up a mass driver on the MooOf course, you can't just take the current market value of platinum, multiply by the amount of platinum available in a large meteorite - or on the Moon if Wingo and Wieczorek et al are right - and conclude that you'd get trillions of dollars by returning all that platinum to Earth and selling it here. You need to fulfill a need or eventually nobody will buy it. If it's just to replace copper, for instance, in wires, it wouldn't be worth returning unless you could reduce the transport cost back to Earth right down. Dennis Wingo suggested in Moonrush that it could be worth exporting it to Earth for use for fuel cells, as an application that could be high value and yet need a lot of platinum.The gold could be useful too, on the Moon at least. You don't normally think of gold as more decorative than useful but it is used a fair bit in electronics Also combined with the abundant titanium on the Moon you get Ti3Au, an alloy with 70% less wear, four times the hardness and increased biocompatibility compared with pure titanium (and twice as hard as titanium / silver and titanium copper alloys). It's also 70% less wear than titanium, lower friction and four times harder with a hardness of 800 HV in the Vickers hardness test. Density about the same as steel.(density of titanium: 4.43 g/cc. using the atomic masses of gold and titanium, multiplying by (196.96657+3*47.867)/(4*47.867)*4.43 = 7.88 approx. By comparison, density of steel is 7.75 g / cc).The paper focuses on its medical applications, you can alloy titanium with copper or silver, which are twice as hard as pure titanium, but this is four times as hard. It's also 70% more resistant to wear which will make it last longer and lead to less debris. And has excellent biocompatibility properties. But I wonder if it might also have lunar applications, with the hardness especially and resistance to wear.Probably only the platinum group metals would be worth returning to Earth, since it's going to be easier to mine the Near Earth Asteroids, especially the ones that consist almost entirely of pure metal. However, whether or not they are useful for Earth, they are well worth using on the lunar surface once you have industry there.The Moon has some advantages over Mars indeed for metals, such as the pure nanophase iron mixed in with the regolith, which can only exist in oxidized form on Mars except for rare metal meteorites. Also, it's unlikely it will be commercially worthwhile to return metals from Mars while there are definite possibilities of returning metals from the Moon. See Exporting materials from the Moon for future suggested low cost methods for export from the Moon. For discussion of whether anything physical could be worth the expense of export from Mars, see Commercial value for MarsLUNAR GLASSThis is a beneficial side effect of all the micrometeorite impacts on the Moon (which you don't get so much on Mars with its thin atmosphere, just enough to filter out micrometeorites). The Moon's "soil" or regolith contains large quantities of glass, created during the impacts. It also has free iron, as we saw, at half of one percent of the soil, in tiny micro beads of iron (nanophase iron) which concentrate the microwave energy. Again, you don't have this on Mars.As a result, it is really fast to melt the regolith using microwaves. It took only 30 seconds to melt small lunar sample at 250 watts (typical of a domestic microwave). You can melt the soil to glass as easily as you can boil water using the microwave in your kitchen. See lunar lawnmower. This only works with genuine lunar soil and not the simulants. We have nothing analogous to lunar soil on Earth, as Larry Taylor, principle author of this paper found: Microwave Sintering of Lunar Soil: Properties, Theory, and Practice. He says the microstructure of the genuine lunar regolith, with nanophase iron beads scattered throughout, would be almost impossible to simulate.His idea (see Products from Microwave Processing of Lunar Soil on page 194 of the paper) is to run a "lunar lawnmower" over the soil with two rows of magnetrons (such as generate microwaves in a microwave cooker). The first row would sinter it to a depth of half a meter using microwaves. Then the second row completely melts the top 3-5 cm of the soil, which then crystallizes to glass. As it does this, it will heat up and release most of the solar wind particles notably hydrogen, helium, carbon and nitrogen. So it could also capture these assets as it goes along, including the Helium 3, if this turns out to be of economic value.See also The Lunar Dust Problem: From Liability to Asset. This could also be useful, for instance, for a solar panel paving robot to make solar panels, and other applications.Then, there's Behrokh Khoshnevis' idea for making a landing pad on the Moon using tiles made of lunar glass in situ. The idea is to make the surface into lots of tiles by injecting a material that can't be sintered easily using microwaves into the soil first to outline the edges of the tiles, then use microwaves to melt the soil in between.This would make a tiled flat surface for supply vessels to land on. It would also help with the problem of lunar dust by removing dust from the landing area. You can read the details here. He used lunar regolith simulant, so presumably by Larry Taylor's results, it would work even better with genuine lunar samples.SOLAR CELLS FROM LUNAR MATERIALS - SOLAR PANEL PAVING ROBOTOnce you have glass, it might not be such a big step to make photovoltaic cells on the Moon. And here the Moon has one big advantage, the high grade vacuum so you could use vacuum deposition to make the cells in situ. To start with you'd make the cells themselves from materials sent from Earth, later on mine them on the Moon.This is a report from the Center for Advanced Materials at the University of Houston, suggesting the possibility of an autonomous solar powered lunar photovoltaic cell production roverIt would use silicon extracted from lunar materials to make the cells themselves. Of the various methods you could use, magma electrolysis may be best. He uses low efficiency silicon cells which are vacuum deposited on glass, something that is not easy to do on Earth but would be possible in the ultra high vacuum conditions on the Moon. Techy details of this suggestion are here.It would require transporting a small mass to the Moon in the form of the rover which then over several years of driving could build a 1 MW facility on the Moon.sIdea for a robot to drive over the surface of the Moon leaving solar panels in its wake wherever it goes, using only indigenous lunar materials to make the panels. The panels would be only 1% efficient, but given that there is no shortage of real estate on the Moon, that might not matter. It might be more important to make the panels in situ without any imports from Earth than to make them highly efficientStructure of the panelsFor making glass on the Moon see the section above: Lunar glassBASALT (LIKE GLASS FIBER)The basalt itself is a natural resource. If reasonably pure and consistent in composition, it's ideal for making basalt fibre, which is like glass wool, but much better in some ways. The regolith consists mainly of powdered basalt. So might well be ideal for making basalt fibre. See:Basalt Fiber PropertiesHELIUM 3I should mention this, since the topic is brought up so often in discussions of lunar settlement. However I don't see this as a major plus point for the Moon at present.The Moon is a source for helium 3, deposited in the regolith by the solar wind, and some say that helium 3 will be of value for fusion power in the future because it is not radioactive and doesn't produce radioactive waste products. If so, small amounts of helium 3 from the Moon could be worth a lot on Earth and be a useful commodity to export. Apollo 17's Harrison Schmidt is a keen advocate of helium 3 mining on at a reasonable rate at a reasonable rate the Moon.However, we don't yet have fusion power plants at all, and one able to use helium 3 is a tougher challenge. Frank Close wrote an article in 2007 describing this idea as "moonshine" saying it wouldn't work anyway. Frank Close says that in a deuterium - helium 3 tokamak, at normal temperatures for a tokamak, the deuterium helium 3 reaction proceeds so slowly that the deuterium would instead fuse with itself producing tritium and then fuse with the tritium (the original article is here, but it's behind a paywall). For a critical discussion see also the Space Review article The helium-3 incantationSee also Mining the Moon by Mark Williams Pontin. If you can use much higher temperatures, six times the temperature at the centre of the sun by some calculations, the helium 3 will fuse at a reasonable rate, but these are temperatures way beyond what is practical in a tokamak at present. The reason such high temperatures are needed for a tokamak is because the plasma is in thermal equilibrium and has a maxwellian distribution which means that to achieve a few particles at very high temperatures you have to heat up a lot of particles to lower temperatures to fill up the maxwellian distribution so that just a few will react. This is potentially feasible for the lower temperatures of DT but not feasible for the higher temperatures of 3He 3He.However if you use electrostatic confinement, a bit like a spherical cathode ray tube with the fusion happening at the center where the negatively charged "virtual cathode" is, then the particles are all at the same high energy and the result is much more feasible with lower power requirements. This is the approach of Gerald Kulcinsky who achieves helium 3 fusion in a reactor 10 cm in diameter. However though it does produce power, it produces only one milliwatt of power for each kW of power input so is a long way from break even at present.Gerald Kulcinski who has developed a small demonstration electrostatic 3He 3He reactor 10 cm in diameter. It is far from break-even at present, producing 1 milliwatt of power output for each kilowatt of input. See A fascinating hour with Gerald KulcinskiPerhaps this line of development will come to something. Perhaps one way or another we will achieve helium 3 fusion as the enthusiasts for helium 3 mining on the Moon hope. However it is early days yet, and we can't yet depend on this based on a future technology that doesn't exist yet.However even if we do achieve helium 3 fusion, it might not be such a game changer for the lunar economy as you might think. Crawford says (page 25) that to supply all of our energy from Helium 3 would mean mining 5000 square kilometers a year on the Moon, which seems ambitious (and would mean the whole Moon would only last 200 years). So, even if we develop Helium 3 based fusion, and it turns out to be a valuable export, it's probably not going to be a major part of the energy mix.Even more telling, he also calculates that covering a given area of the Moon with solar panels would generate as much energy in 7 years as you'd get from extracting all the Helium 3 from that region to a depth of three meters.Also - there are many other ideas being developed for nuclear fusion, such as laser fusion, and the polywell which has the same advantage that no signNow radiation is produced when it uses fusion of boron and hydrogen. I think it is far too soon to know whether or not the helium 3 on the Moon will be an asset in the future when we achieve nuclear fusion power. For a summary, see ESA: Helium-3 mining on the lunar surface.This doesn't mean that there is no point in helium 3 mining however. As Crawford suggests (page 26), Helium 3 is useful for other things, not just for fusion power. It's used for cryogenics, neutron detection, and MRI scanners, amongst other applications, so some Helium 3 from the Moon could be a valuable export right away, even if it doesn't scale up to the huge quantities you'd need for Helium 3 based power generation on Earth. You'd get it automatically as a byproduct while extracting the more abundant volatiles from the solar wind in the regolith, so it might well be a useful side-line to help support lunar manufacturing economically as part of the mix along with everything else.THORIUM AND KREEP (POTASSIUM, PHOSPHORUS AND RARE EARTH ELEMENTS) ,AND SOME URANIUMThe Moon has some uranium, which is a bit of a surprise for such a heavy element, but when bound with oxygen it is rather lighter and can occur in the lunar crust as on Earth. It is especially rich in Thorium, in the lunar Mare. This is useful as a fuel for nuclear fission reactors, which have to be designed to burn thorium instead of uranium to use it. It's not likely to be worth returning to Earth as thorium is abundant here. But it could be very useful in space, at some point in the future.Nuclear power stations built on the Moon wouldn't have the same pollution hazards and hazardous waste issues as stations on the Earth. Perhaps this may be a way to power space colonies, and interplanetary ships fueled from the Moon, so avoiding the need to launch nuclear power plants from Earth to orbit.Thorium is a tracer for KREEP - potassium, phosphorus and rare earth elements. Also associated with chlorine, fluorine, sodium, uranium, thorium, and zirconium, so KREEP ores could be sources for all those elements on the Moon.When the Moon cooled down from the original molten state, then olivine and pyroxene crystals form first, and sink to the bottom of the magma ocean (both made of iron and/or magnesium plus silicon and oxygen). Meanwhile anorthite also forms (made of calcium, aluminum, silicon, and oxygen), which is less dense and floats to the top (forming the lunar highlands). Some of the other elements like nickel are able to squeeze into the crystal lattice and get removed at the same time. But the larger elements can't, and are left in liquid state. They are last to solidify and form the KREEP deposits. It forms in between the olivine and pyroxene deep down, and the floating anorthite on top and may have been liquid for a long time.For some reason, not fully understood, then KREEP deposits on the surface of the Moon are concentrated on the near side of the Moon near the Imbrium basin, with a small amount also in a separate concentration on the far side. The Imbrium impactor probably excavated the KREEP deposits on the near side. But it's puzzling that the much larger Aitken basin didn't lead to large deposits on the far side. Perhaps for some reason KREEP is concentrated on the near side of the Moon. For more about this see The Moon is a KREEPy place by the planetary geologist Emily Lakdwalla which I summarized here.The abundances of rare earth elements on the Moon are much less than rare earth ores on Earth, and despite the name, they aren't very rare here on Earth. So it's not likely that they'll be worth returning. However the most concentrated spots - the ones marked white in this figure - haven't been sampled on the surface and the spatial resolution is low, tens of kilometers. So it's possible we'll find more concentrated ores on the Moon.It's a similar situation for uranium and thorium. The abundances on the Moon from this map are too low to count even as a low grade ore on Earth. But with such low resolution, there could be richer ore deposits when we look at it closely. (Here I'm summarizing what Crawford says about lunar KREEP ores in his survey, see section 7, Rare earth elements and following)POSSIBILITY OF USING LUNAR SOLAR POWER FOR EARTHThis is a bit further ahead, but it is worth thinking about, whether solar power for the Moon could actually be useful for Earth also. Some scientists think it could be.The advantage of doing this on the Moon is that you can use indigenous materials to make the solar panels. For a small amount of launch mass to the Moon you could have a rover that travels over the surface leaving solar panels in its wake. See Lunar glass and Solar cells from lunar materials - solar panel paving robot (above)It's easy to see this working to supply power to the Moon, but some have suggested it could also be used to generate power on Earth. So, taking this even further, with a large scale operation of this type, using only 1% of the surface area of the Moon, you could supply 2 kilowatts of continuous power per person to a population of 10 billion on the Earth. See Solar Power via the Moon. More details here.Or, further ahead, maybe this is more interesting as a talking point than a likely near future concept, the Japanese Shingzu corporation has suggested we could build solar panels in a band around the Moon - at the equatorSee Shimizu dream - Lunar Solar Power Generation - Luna Ring.Earth would get solar power only half the day, so they send the power to satellites in orbit around Earth, which then beam it down to the other side of Earth. Of course they need large receivers to collect the power from the Moon, but only 1% of what they'd need to collect it directly from the sun - that could be worth doing if it is signNowly easier to make solar panels on the Moon.On the other hand there are ideas to use large thin film solar panels in space or large thin film mirrors to concentrate the light onto solar panels or furnaces, launched from Earth to LEO. So would the lunar solar plants be a major saving compared to those?Another way that the Moon could help the Earth though, with solar power, is to make the solar cells from lunar materials, and then ship them to GEO or lower orbit. The idea of using lunar materials to make solar power satellites goes back at least to the 1970s, see Construction of Satellite Solar Power Stations from Nonterrestrial MaterialsFor more on this see the The Moon is resource rich section of my Case for Moon FirstWhether it is useful off planet depends a lot on how easy it is to export the materials from the Moon, and one of the most promising ways to do that is Hoyt’s cislunar tether system which exploits the Moon’s position as higher in the gravitational well than Earth to basically “roll the goods down hill” from the Moon to Earth through a system of rotating tethers.SeeExporting materials from the MoonBallutes - return of high value resources such as platinum to Earthin my Case For Moon First
-
What is the Reliable Dollar Colony?
Reliaable Dollar Colony-Reliaable Dollar Colony is developers. This coming soon project is located on the point of HSR layout, off Hosa Road in Bangalore. The first ten acres of the open, sixty acres BDA approved best quality development is put to launch in March 2017. Phase I will proffer 115 plots put within ten acres of landscaped region with a crowd of modern facilities. Motivated by Bangalore's old world charm by way of sprawling regions in the lap of luxury, thisReliaable single among the best major BDA Approved Gated Community scheme in Bangalore yet planned by highly modern project promises to propose luxury living with an existing lifestyle.Located at the best destination of Avalahalli Off Sarjapur Road is a suburban region situated in the western element of Bangalore in Karnataka. It is positioned on the point of one of the highly famous temples of theVimetropolis, the Gavi Gangadhareshwara Temple. Here, 60 acres of gated development region containing of 115 luxuries Villa plots at extremely reasonable rates. Yes it is Reliaable Dollars Colony Hosa Road brought of Hosa Road.very tactical location by at builder reliableThe locality is bordered by Ganapathi Nagar, Srinagar, New BDA Layout, Telecom Colony, Muneshwara Block along with Girinagar. Close nearness to all facilities and the tranquil environment of the region has made it a preferential housing destination in Bangalore.Some of the most important roads of Bangalore are positioned close to Avalahalli off Sarjapur Road together with the Road, Outer Ring Road along with the NICE Expressway. BMTC Buses as well serve the neighborhood and the stops positioned close by greeneries. Metro service has also been projected in the metropolis that would ease traffic in the future.Avalahalli off Sarjapur Road is situated 5.5 kilometers from the City Junction Railway Station and can be attained within 15 minutes via the Tank Bund Road. The HAL International Airport is only just 30- minute drive away from this Road.If we talk about Educational Institutions, Max Muller High School, Kidigos Play School, SGM Public School, Jnana Deepthi English School and Ajji Mane School are a few of the admired schools approximately Avalahalli Off Sarjapur Road.Famous colleges near the area of this project Reliaable Dollars Colony Hosa Road Bangalore comprise of Engineering.B College and gement Medical College, NRI Institutes, Osteen College of rajeshwari PES College of Pharmacy,Owing to its nearness to foremost attractions of the metropolis and well-developed transport network, Avalahalli has appeared as a preferred residential position. The area is drawing the concentration of numerous prestigious developers as Projects, Builders, Group, SJR Primecorp, Builders in addition to Pioneer Developers who have started their projects comprising plots, apartments in addition to villas.A number of well-liked constant projects in the region are Hamilton Homes by SJR Primecorp, Gardenia by famous Builders, Ample by Ambience Housing Projects and Verdure by Projects. Developed region of apartments in Avalahalli is ranging between 466 sq. ft. as well as Rs. 2800 per sq. ft., in that order.ft. with the utmost and lowest amount prices being Rs. 5990 per sq. ft. The average real estate price in the region is Rs.3588 per sq. ft. and 5150 sq.Builder-Reliaable Developers are familiar as one of the highly prolific and outstanding property developers in Bangalore. Since beginning in 1999, they have added an alarming presence in the whole Bangalore that can be qualified to our sustained deliverance in present residence owners their idyllic living region. They have enlarged both specially selected urban and luxury living regions in over crosswise 1000 acres. The company was industrial with an assignment to make easy generous and profitable real estate investment we have delivered schemes that have undergone hundred times growth of extending 10,000 BDA approved plots that square feet cost. They offer steady high standards and hunt for superiority in an exceedingly aggressive.marketSignpost projects of Reliaable Avia in Devanahalli, and so on.Reliaable Woods in HSR Layout, Tranquil in Harlur, Silver Oak in HSR Layout, Elegaance in Off Sarjapur Road, Reliaable in Avalahalli Off Sarjapur Road, Lifestyle in Kasavanahalli Off Sarjapur Road, Magnoliaa in Avalahalli Off Sarjapur Road, Lake Vista in Harlur, The Signature At South in Avalahalli Off Sarjapur Road, Lavendula in Electronic City Phase 2, Gardeniaa in Reliaable Electronic City Phase 2, Sunfields Developers are Acacia in Bellandur, Lakedew Residency in Harlur,
Trusted esignature solution— what our customers are saying
Get legally-binding signatures now!
Related searches to Draw Electronic signature Presentation Easy
Frequently asked questions
How do i add an electronic signature to a word document?
How to create electronic signature image?
Pdf element how to sign document on phone?
Get more for Draw Electronic signature Presentation Easy
- How To Electronic signature Texas Police PDF
- How To Electronic signature Texas Police Word
- How Do I Electronic signature Texas Police Word
- How Do I Electronic signature Texas Police Word
- How Do I Electronic signature Texas Police PDF
- Help Me With Electronic signature Texas Police Word
- Help Me With Electronic signature Texas Police Word
- How Can I Electronic signature Texas Police Word
Find out other Draw Electronic signature Presentation Easy
- Unconditional waiver final form
- Agreed written termination of lease by landlord and tenant oregon form
- Demand by mortgagee for list of labor and materials individual oregon form
- Notice of breach of written lease for violating specific provisions of lease with right to cure for residential property from 497323748 form
- Notice breach lease 497323749 form
- Insurers report oregon form
- Notice of breach of written lease for violating specific provisions of lease with no right to cure for residential property 497323751 form
- Insurer notice of closure summary oregon form
- Oregon provisions form
- Vocational reimbursement request oregon form
- List labor form
- Report of gross annual income oregon form
- Notice of closure oregon form
- Oregon notice form
- Permanent total disability form
- Oregon notice closure form
- Oregon notice 497323763 form
- Workers compensation surety rider oregon form
- Oregon entity form
- Oregon assistance form